Market Trends
Key Emerging Trends in the Pyrogen Testing Market
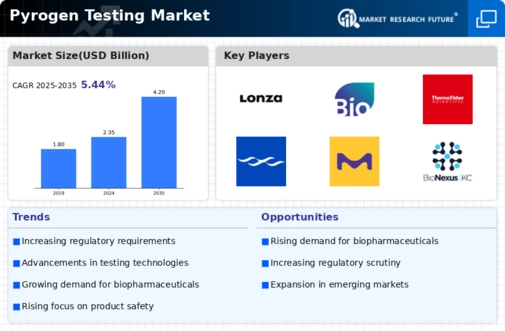
The Pyrogen Testing marketplace is gaining prominence, driven by the growing importance of ensuring the safety of pharmaceutical and clinical tool products. Pyrogen Testing performs a critical function in detecting fever-inducing contaminants, which include endotoxins, which could have severe fitness implications if found in clinical merchandise. Stringent regulatory standards, especially within the pharmaceutical and scientific device sectors, are propelling the adoption of Pyrogen Testing. Compliance with those standards is imperative for product approval, creating a robust demand for dependable checking-out techniques inside the market. The enterprise's increasing recognition of endotoxin-unfastened production methods is boosting the demand for Pyrogen Testing. Manufacturers are incorporating strong testing protocols to make certain that their merchandise is free from pyrogenic contaminants, aligning with the emphasis on patient safety. The globalization of pharmaceutical production is contributing to the growth of the Pyrogen Testing marketplace. As manufacturing centers spread globally, the need for standardized and reliable Pyrogen Testing will become crucial to maintain product quality and protection. The market is witnessing a growing demand for automated Pyrogen Testing answers. Automated systems provide performance, accuracy, and reduced hazard of contamination throughout the testing manner, making them attractive to industries searching for streamlined manufacturing strategies. Advancements in opportunity testing methods, together with monocyte activation exams (MAT) and in vitro Pyrogen Testing, are gaining interest. These strategies offer extra options for Pyrogen Testing, imparting alternatives to traditional LAL assays and expanding the market's technological landscape. Despite development, demanding situations in looking at standardization persist. Harmonizing Pyrogen Testing strategies to ensure consistency across specific products and manufacturers creates an ongoing awareness for the enterprise to facilitate smoother regulatory compliance. Education and schooling projects play a critical function in increasing the market. Training packages and workshops centered on Pyrogen Testing methods and nice practices are helping industry experts stay updated on present-day improvements and compliance requirements. Collaborations and partnerships between corporations are shaping the aggressive landscape of the Pyrogen Testing market. Such strategic alliances are facilitating the improvement of completely trying out answers and increasing the reach of those technologies.



















Leave a Comment