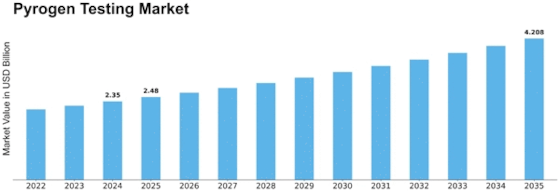
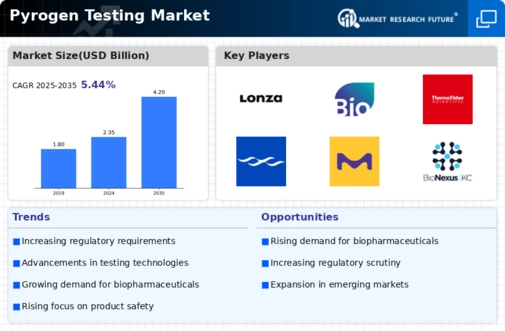
Pyrogen Testing Size
Pyrogen Testing Market Growth Projections and Opportunities
The Pyrogen Testing marketplace is drastically encouraged by the increase in the pharmaceutical and biotechnology industries. As these sectors become bigger globally, the demand for rigorous testing of pharmaceutical products to ensure certain freedom from pyrogens becomes essential, driving market growth. The globalization of pharmaceutical production ends in accelerated cross-border exchange and collaboration. Pyrogen testing becomes essential in ensuring the protection and best of pharmaceutical products, fostering growth in the marketplace. Ongoing advancements in Pyrogen Testing strategies contribute to marketplace evolution. Innovative and efficient checking out technologies, which include the Limulus Amebocyte Lysate (LAL) test, decorate sensitivity, lessen testing time, and improve accuracy, impacting market dynamics. Endotoxin testing, a subset of Pyrogen Testing, is witnessing accelerated emphasis because of its relevance in excellent pharmaceutical control. The market is pushed via the growing recognition of the effect of endotoxins on affected persons' health and the want for reliable trying-out solutions. The prevalence of pyrogen infection in pharmaceutical merchandise and medical devices maintains an upward push. This drives the demand for effective Pyrogen Testing answers as groups strive to fulfill high-quality standards and avoid regulatory issues. The Pyrogen Testing market is stimulated by focus and schooling initiatives regarding the dangers associated with pyrogen infection. Efforts to train experts within the pharmaceutical and biotechnology industries contribute to improved adoption and marketplace growth. Collaborations and partnerships between Pyrogen Testing solution providers and pharmaceutical companies influence marketplace trends. These collaborations frequently result in the development of superior trying-out solutions tailored to enterprise needs, impacting adoption quotes. The price-effectiveness of Pyrogen Testing answers is a critical factor influencing marketplace dynamics. Affordable and green testing methods appeal to a broader variety of users, including smaller pharmaceutical businesses and research laboratories. Efforts to validate and standardize Pyrogen Testing strategies increase contributions to the marketplace. The established order of standardized protocols enhances the reliability and comparison of check outcomes, promoting the adoption of Pyrogen Testing across the industry. Emerging markets with growing pharmaceutical and biotechnology sectors gift untapped possibilities for the Pyrogen Testing marketplace.



















Leave a Comment