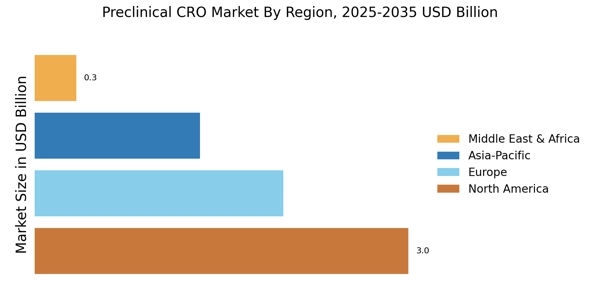
The Preclinical CRO Market size is currently experiencing a dynamic evolution, driven by the increasing demand for efficient drug development processes. As pharmaceutical and biotechnology companies seek to streamline their research and development efforts, the reliance on Contract Research Organizations (CROs) has intensified. These organizations provide essential services, including toxicology studies, pharmacokinetics, and efficacy testing, which are crucial in the early stages of drug discovery. The growing complexity of regulatory requirements further propels the need for specialized expertise, making preclinical CROs indispensable partners in the drug development landscape.
Moreover, advancements in technology, such as the integration of artificial intelligence and data analytics, are reshaping the operational frameworks of preclinical CROs. These innovations enhance the accuracy and efficiency of preclinical studies, allowing for more informed decision-making. As the market continues to expand, collaboration between CROs and pharmaceutical companies is likely to deepen, fostering a more integrated approach to drug development. This trend suggests a shift towards a more collaborative ecosystem, where shared knowledge and resources can lead to accelerated timelines and improved outcomes in the quest for new therapeutics.
Technological Integration
The Preclinical CRO Market is witnessing a notable trend towards the integration of advanced technologies. Innovations such as artificial intelligence and machine learning are being adopted to enhance data analysis and streamline research processes. This technological shift not only improves the efficiency of preclinical studies but also allows for more precise predictions regarding drug efficacy and safety.
According to the World Health Organization, digital health and advanced analytics adoption is accelerating globally, with over 120 countries implementing digital health strategies, enabling the integration of AI and data-driven tools in healthcare research. Additionally, the Centers for Disease Control and Prevention is actively expanding advanced data analytics and modeling systems for disease monitoring, while insights from the Institute for Health Metrics and Evaluation highlight the increasing use of large-scale health data to improve predictive modeling and clinical research efficiency.
Regulatory Compliance Focus
There is an increasing emphasis on regulatory compliance within the Preclinical CRO Market. As drug development becomes more complex, CROs are prioritizing adherence to stringent regulatory standards. This focus ensures that studies are conducted in accordance with guidelines, thereby reducing the risk of delays in the approval process and enhancing the credibility of research outcomes.
The World Health Organization reports that over 60% of countries have established regulatory frameworks or policies for health data governance and clinical research standards, supporting safer and more standardized drug development processes. Furthermore, the European Centre for Disease Prevention and Control emphasizes strengthening regulatory and surveillance systems across Europe to ensure compliance and improve public health outcomes, reinforcing the importance of adherence in preclinical and clinical research.
Collaborative Partnerships
The trend of forming collaborative partnerships is gaining traction in the Preclinical CRO Market. Pharmaceutical companies are increasingly seeking long-term relationships with CROs to leverage their expertise and resources. Such collaborations facilitate knowledge sharing and foster innovation, ultimately leading to more efficient drug development processes.
The World Health Organization reports that over 60% of countries have established regulatory frameworks or policies for health data governance and clinical research standards, supporting safer and more standardized drug development processes. Furthermore, the European Centre for Disease Prevention and Control emphasizes strengthening regulatory and surveillance systems across Europe to ensure compliance and improve public health outcomes, reinforcing the importance of adherence in preclinical and clinical research.