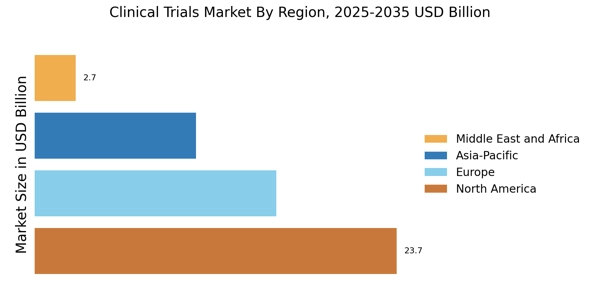
North America : Clinical Trials Market Powerhouse
North America led the market with over 40% share, generating around USD 23.7 billion in revenue. Key growth drivers include advanced healthcare infrastructure, high R&D investments, and a robust regulatory framework that encourages innovation. The U.S. is the largest market, followed by Canada, which holds around 10% of the market share. Regulatory catalysts, such as expedited approval processes, further enhance the region's attractiveness for clinical research.
The competitive landscape in North America is characterized by the presence of major players like IQVIA, Covance, and PPD, which contribute significantly to the market's growth. The U.S. is home to numerous clinical trial sites, supported by a diverse patient population and a strong emphasis on patient-centric research. This environment fosters collaboration among stakeholders, including pharmaceutical companies, CROs, and regulatory bodies, ensuring a steady pipeline of clinical trials and innovations.
Europe : Emerging Clinical Trials Market Hub
Europe Clinical Trials Market size was valued at USD 15.834 billion in 2024, making it the second-largest regional market with a 30% share. The region benefits from a harmonized regulatory environment, particularly through the European Medicines Agency (EMA), which streamlines the approval process for clinical trials. Countries like Germany and the UK are leading the market, with Germany holding about 12% of the share, driven by strong healthcare systems and increasing investments in biopharmaceutical research.
The competitive landscape in Europe features a mix of local and international players, including Covance and Charles River Laboratories. The presence of diverse patient populations across member states enhances recruitment for clinical trials. Additionally, the region's focus on innovative therapies and personalized medicine is attracting more clinical research activities, making Europe a key player in The Clinical Trials Market.
Asia-Pacific : Rapidly Growing Market
Asia-Pacific is rapidly emerging as a significant player in the clinical trials market, accounting for approximately 20% of the global share. The region's growth is driven by increasing healthcare expenditures, a large patient population, and favorable regulatory environments in countries like China and India. China is the largest market in the region, holding about 10% of the global share, supported by government initiatives to boost clinical research and development. The competitive landscape in Asia-Pacific is evolving, with both local and international CROs expanding their presence.
Key players like Wuxi AppTec and KCR are capitalizing on the region's growth potential. The increasing focus on innovative therapies and the rising demand for cost-effective clinical trials are further propelling the market. Collaborative efforts between governments and private sectors are also enhancing the region's capabilities in conducting clinical trials.
Middle East and Africa : Emerging Clinical Trials Market Landscape
The Middle East and Africa region is gradually emerging in the clinical trials market, holding approximately 5% of the global share. Growth is driven by increasing investments in healthcare infrastructure and a rising number of clinical research organizations. Countries like South Africa and the UAE are leading the market, with South Africa accounting for a significant portion due to its diverse patient population and established regulatory frameworks that support clinical trials.
The competitive landscape in this region is characterized by a growing number of local CROs and partnerships with international firms. The presence of key players is increasing, with a focus on enhancing clinical trial capabilities. Additionally, government initiatives aimed at improving healthcare access and regulatory support are fostering a more conducive environment for clinical research, making the region an attractive destination for clinical trials.