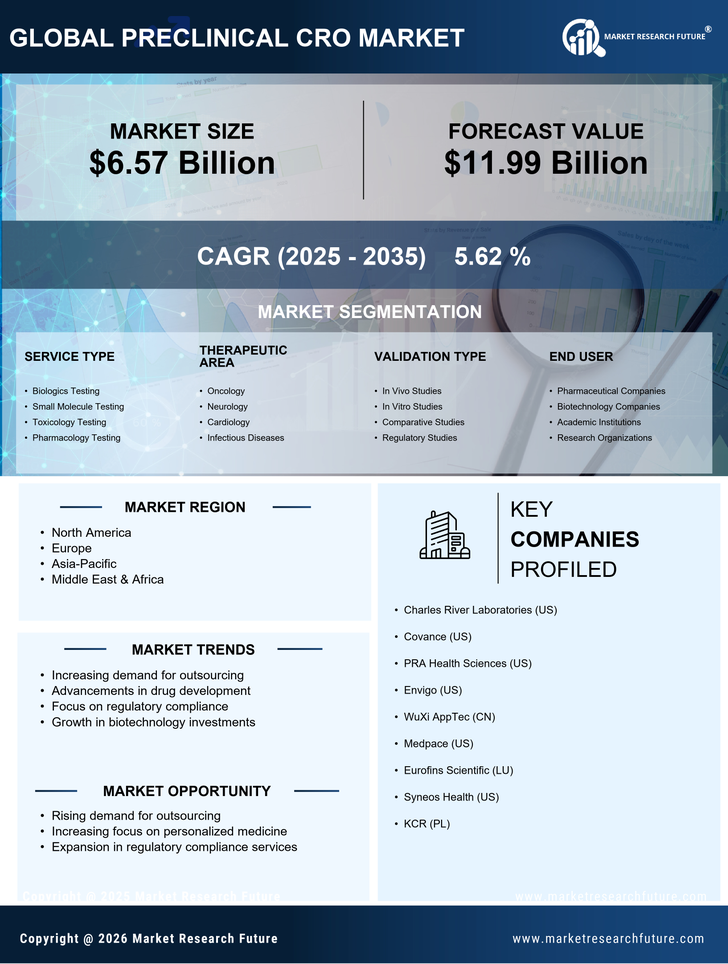
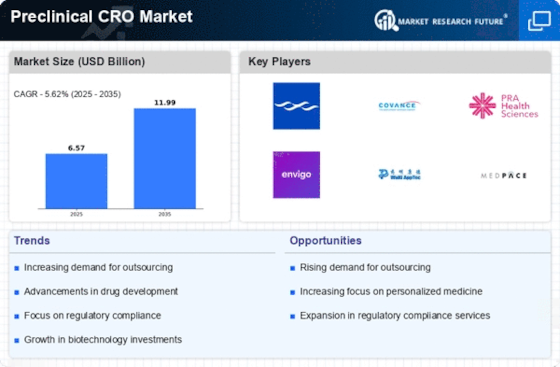
Healthcare Market Segmentation
Healthcare By Service Type (USD Billion, 2025-2035)
- Biologics Testing
- Small Molecule Testing
- Toxicology Testing
- Pharmacology Testing
Healthcare By Therapeutic Area (USD Billion, 2025-2035)
- Oncology
- Neurology
- Cardiology
- Infectious Diseases
Healthcare By Validation Type (USD Billion, 2025-2035)
- In Vivo Studies
- In Vitro Studies
- Comparative Studies
- Regulatory Studies
Healthcare By End User (USD Billion, 2025-2035)
- Pharmaceutical Companies
- Biotechnology Companies
- Academic Institutions
- Research Organizations