Blood Screening Market Summary
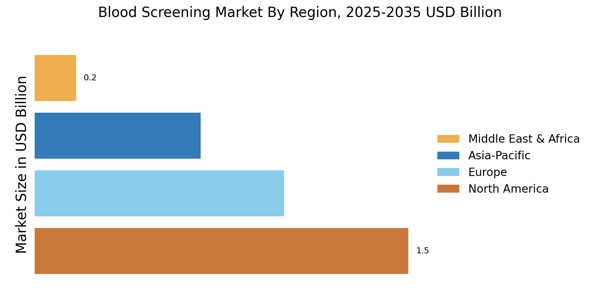
As per MRFR analysis, the Blood Screening Market size was valued at USD 3.4 Billion in 2024. The market is projected to grow from USD 3.747 Billion in 2025 to USD 9.898 Billion by 2035, exhibiting a CAGR of 10% during the forecast period 2025-2035. North America led the market with over 45% share, generating around USD 1.5 Billion in revenue.
The Blood Screening Market is expanding due to increasing prevalence of infectious and chronic diseases, rising demand for early disease detection, and growing adoption of advanced screening technologies. Key trends include integration of automated and rapid diagnostic platforms, expanding blood safety programs, and rising government initiatives to enhance healthcare screening infrastructure globally.
According to the World Health Organization, screening for diseases such as HIV, hepatitis B, and hepatitis C is critical to reduce transmission and improve public health outcomes.
Key Market Trends & Highlights
The Blood Screening Market is poised for substantial growth driven by technological advancements and increasing health awareness.
- Technological advancements in blood screening are enhancing diagnostic accuracy and efficiency, particularly in North America.
- The Asia-Pacific region is emerging as the fastest-growing market, fueled by rising healthcare investments and infrastructure improvements.
- Nucleic Acid Amplification Tests dominate the market, while Next-Generation Sequencing is rapidly gaining traction as a preferred method.
- Key market drivers include the increasing prevalence of infectious diseases and rising awareness of blood donation and screening initiatives.
Market Size & Forecast
| 2024 Market Size | 3.4 (USD Billion) |
| 2035 Market Size | 9.898 (USD Billion) |
| CAGR (2025 - 2035) | 10.2% |
Major Players
Companies such as Abbott Laboratories (US), Roche Diagnostics (CH), Siemens Healthineers (DE), Thermo Fisher Scientific (US), Becton Dickinson and Company (US), Ortho Clinical Diagnostics (US), Grifols (ES), Bio-Rad Laboratories (US), Hologic (US) are some of the major participants in the global market.