全外显子组测序市场概览
p根据 MRFR 分析,2024 年全外显子组测序市场规模估计为 21.6 亿美元。预计到 2034 年,全外显子组测序市场规模将从 2025 年的 26.0 亿美元增长到 136.2 亿美元,预测期内(2025 - 2034 年)的复合年增长率 (CAGR) 预计约为 20.20%。全外显子组测序技术在各种应用和研究中的使用日益增多。发展是促进市场增长的关键市场驱动力。
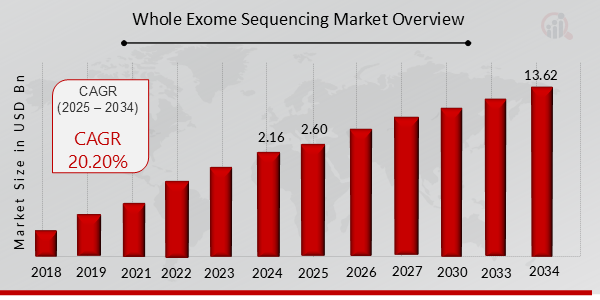
来源二手研究、一手研究、MRFR 数据库和分析师评论
全外显子组测序市场趋势
h3
全外显子组测序技术的日益普及正在推动市场增长 p全外显子组测序技术各种应用的市场复合年增长率受到慢性和罕见疾病数量的增加、对定制治疗的需求的增加以及下一代测序的兴起的推动。对基因组的外显子组和蛋白质编码基因进行测序的方法称为外显子组测序和全外显子组测序。在此过程中,选择编码蛋白质或外显子的DNA子集,然后利用高通量DNA测序技术对外显子DNA进行测序。此外,它还提供了一种用于检测遗传疾病的低成本筛选方法。它为包括药物开发过程在内的多种应用提供了一种资源密集度更低的全基因组测序替代方案。
对识别罕见疾病的需求日益增长、基因组学和下一代测序研发的不断扩展以及对个性化治疗的需求不断增长,推动了整个外显子组测序市场的发展。例如,根据2022年3月的《英国医学杂志》文章,现在已为经过严格筛选的儿童提供用于检测罕见儿童遗传疾病的常规全外显子组测序。根据该报告,下一代测序能够快速且廉价地对数百或数千个基因进行测序。外显子组测序的优势预计将是推动市场增长的因素。
检测导致HIV、癌症和Covid-19等疾病的病毒基因组涉及全外显子组测序。随着这些疾病的流行,对全外显子组测序的需求也在迅速增长。例如,世界卫生组织 (WHO) 于 2022 年 7 月宣布,2021 年全球有 3770 万例艾滋病毒病例。由于这些疾病的增加以及对 RNA 测序需求的增加,这些基因组测序技术提供了可能导致疾病的基因变异的信息。从而推动了全外显子组测序市场的收入。
全外显子组测序市场细分洞察
h3
全外显子组测序产品洞察 p基于产品的全外显子组测序市场细分包括试剂盒、系统和服务。系统细分市场占据了市场主导地位,占据了最大的市场收入。下一代测序 (NGS) 和数据分析服务提供了先进的生物信息学可视化,使研究人员能够更加专注于他们的工作,而不是费力的分析操作。这些序列分析服务提供各种服务,例如从各种样本中构建文库、实验规划以及下游 DNA 序列分析。
全外显子组测序技术洞察
p基于技术的全外显子组测序市场细分包括边合成边测序 (SBS) 和 ION 半导体测序。边合成边测序 (SBS) 细分市场的收入最高。该技术通过重复核苷酸序列来消除错误和漏判。SBS 技术除了高分辨率基因组测序外,还提供用于从头测序、有效序列组装和其他用途的长插入双端读取和短插入双端读取。
全外显子组测序应用洞察
p基于应用的全外显子组测序市场细分包括诊断、药物发现和开发以及农业和动物研究。药物发现和开发细分市场的收入最高。得益于遗传学的进步,医学专家可以为各种健康问题制定更安全、更有效的治疗方案和药物。这一药物研发细分市场的发展受到多种因素的推动:各种
癌症患病率的上升、个性化药物治疗的可负担性、发达市场的广泛采用以及新型药物的开发。全外显子组测序最终用户洞察
p全外显子组测序市场细分基于最终用户,包括研究中心和政府机构、医院和诊断中心以及制药和生物技术公司。研究中心和政府机构类别的收入最高。研究中心和政府机构提供心血管疾病、阿尔茨海默病、癌症等慢性疾病的临床诊断。此外,研发基因组学领域的研发也支撑了该领域的增长。
全外显子组测序区域洞察
p按区域划分,本研究提供了北美、欧洲、亚太地区和世界其他地区的市场洞察。由于遗传疾病和慢性疾病的增加,北美全外显子组测序市场将占据主导地位。此外,对靶向和个性化药物的不断增长的需求将推动该地区的市场增长。
此外,市场报告中研究的主要国家是美国、加拿大、德国、法国、英国、意大利、西班牙、中国、日本、印度、澳大利亚、韩国和巴西。
图 2 2022 年全外显子组测序市场各地区份额(十亿美元)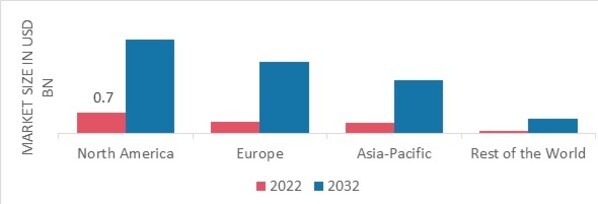
来源:二手研究、一手研究、MRFR 数据库和分析师评论
欧洲全外显子组测序市场占由于基因组学领域的技术进步和研发投入的增加,德国占据了第二大市场份额。此外,德国全外显子组测序市场占有最大的市场份额,英国全外显子组测序市场是欧洲地区增长最快的市场。
由于公众对全外显子组测序的认识不断提高,导致人群中基因异常的发病率不断上升,预计亚太地区全外显子组测序市场将在 2023 年至 2034 年期间以最快的复合年增长率增长。此外,中国的全外显子组测序市场占有最大的市场份额,印度全外显子组测序市场是亚太地区增长最快的市场。
全外显子组测序主要市场参与者和竞争洞察
p领先的市场参与者正在大力投资研发以扩展其产品线,这将有助于全外显子组测序市场进一步增长。市场参与者也在开展多项战略活动来扩大其足迹,重要的市场发展包括新产品发布、合同协议、并购、增加投资以及与其他组织的合作。整个外显子组测序行业必须提供具有成本效益的产品,才能在竞争更激烈、不断增长的市场环境中扩张和生存。
本地化生产以最大限度地降低运营成本是全外显子组测序行业制造商用来造福客户和扩大市场部门的关键商业策略之一。近年来,全外显子组测序行业提供了一些最重要的医疗优势。全外显子组测序市场的主要参与者包括 Illumina Inc.(美国)、BGI(中国)、Eurofins Scientific(比利时)、Thermo Fisher Scientific(美国)、Agilent Technologies Inc.(美国)、F.Hoffmann-La Roche Ltd.(瑞士)、GENEWIZ(美国)、Ambry Genetics(美国)、Macrogen Inc.(韩国)、Integragen SA(法国)等,他们正试图通过投资研发业务来增加市场需求。
Illumina, Inc.(美国)是一家总部位于加利福尼亚州圣地亚哥的生物技术公司,为 140 多个国家/地区提供服务。Illumina 成立于 1998 年 4 月 1 日,致力于创建、生产和销售用于研究遗传变异和生物功能的集成系统。该公司提供一系列产品和服务,以满足测序、基因分型和基因表达以及蛋白质组学市场的需求。例如,2023年2月,Illumina公司向布罗德研究所(Broad Institute)提供了首台NovaSeqX Plus测序仪。该平台将帮助希望访问该公司人类全基因组产品、外显子基因组/外显子组产品和测序服务的各方。NanoString Technologies, Inc.是一家上市生物科技公司,提供发现和转化研究解决方案。nCounter®基因表达系统、GeoMx®数字空间分析器、CosMxTM空间分子成像仪和AtoMX空间信息学平台是NanoString的部分产品。借助这四个系统,研究人员可以三维观察分子相互作用,并了解基因和蛋白质在组织形态的多基因组水平上的表达方式。NanoString的产品基于系统生物学研究所的Leroy Hood博士开发的尖端数字分子条形码技术。例如,2022 年 5 月,NanoString Tehnologies, Inc. 发布了一项基于云的流程,使 GeoMx Digital Spatial Profiler 以及 Illumina NextSeq 1000 和 NextSeq 2000 测序设备的用户受益。
全外显子组测序市场的主要公司包括
ul
Illumina Inc.(美国) 华大基因(中国) Eurofins Scientific(比利时) 赛默飞世尔科技(美国) 安捷伦科技公司(美国) 霍夫曼-罗氏有限公司(瑞士) GENEWIZ(美国) Ambry Genetics(美国) Macrogen Inc.(韩国) Integragen SA(法国) h2
全外显子组测序行业发展 p近期的政策决策,包括与中国的贸易战以及科研经费削减,使Illumina面临重大挑战。中国为报复新关税,将Illumina列为“不可靠实体”,进一步加剧了这些问题。在美国国内,美国国立卫生研究院(NIH)对研发费用的新上限也给该公司带来了更大的压力。此外,市场竞争加剧,例如罗氏公司推出新款测序仪,进一步削弱了Illumina的市场地位。因此,Illumina 的股价今年下跌了 25% 以上,过去五年下跌了近 70%,反映出其在激烈的竞争和监管障碍下处于弱势。尽管遭遇这些挫折,Illumina 仍然通过专注于创新和新兴基因组学市场保持了其主导的市场份额。
2022 年 5 月 赛默飞世尔科技与卡塔尔基金会 (QF) 下属的卡塔尔基因组计划 (QGP) 携手合作,共同推动卡塔尔的基因组研究和预测基因组学的治疗应用。
2021 年 2 月 Labcorp 和 OmniSeq(一家获得 CAP 认证的 Rosewell Park 综合癌症中心的分子诊断创新公司)推出了 OmniSeq INSIGHTsm,这是一项基于组织的全面基因组和免疫学分析测试,结合了下一代测序 (NGS) 技术。 OmniSeq 是 Rosewell Park 综合癌症中心经 CAP 认证的分子诊断发明。
全外显子组测序市场细分
h3
全外显子组测序产品展望 ul
试剂盒 测序仪 服务 h3
全外显子组测序技术展望 ul
边合成边测序 (SBS) ION 半导体测序 h3
全外显子组测序应用展望 ul
诊断 药物发现和发展
农业和动物研究 h3
全外显子组测序最终用户展望 ul
研究中心和政府机构 医院和诊断中心 制药和生物技术公司 h3
全外显子组测序区域展望 p
北美 p
欧洲 p
亚太地区 - 中国
- 日本
- 印度
- 澳大利亚
- 韩国
- 澳大利亚
- 亚太其他地区
p
世界其他地区















