Orphan Drugs Market Summary
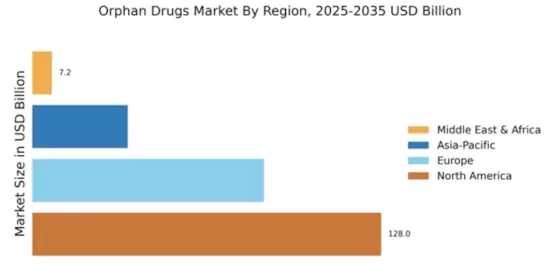
According to MRFR analysis, the Orphan Drugs Market Size was valued at USD 255.18 Billion in 2024. The Orphan Drugs industry is projected to grow from USD 278.35 Billion in 2025 to USD 663.94 Billion by 2035, registering a CAGR of 9.08% during the forecast period (2025–2035). North America led the market with over 50.16% share, generating around USD 128 billion in revenue.
The Orphan Drugs Market is expanding due to increasing focus on rare disease treatments and supportive regulatory incentives. Key trends include advancements in targeted therapies, growing investment in gene and cell therapies, and rising collaboration between pharmaceutical companies and research institutions to accelerate development and improve access to treatments for rare conditions globally. Data from the Institute for Health Metrics and Evaluation indicates that rare and genetic disorders contribute significantly to long-term disease burden, supporting increased investment in orphan drug development.
Key Market Trends & Highlights
The Orphan Drugs Market is experiencing robust growth driven by regulatory support and technological advancements.
- North America remains the largest market for orphan drugs, fueled by strong regulatory incentives and patient advocacy.
- The Asia-Pacific region is emerging as the fastest-growing market, driven by increasing investments in rare disease research.
- Oncology continues to dominate the orphan drugs segment, while genetic disorders are witnessing the fastest growth due to heightened awareness.
- Rising prevalence of rare diseases and advancements in biotechnology are key drivers propelling the orphan drugs market forward.
Market Size & Forecast
| 2024 Market Size | 255.18 (USD Billion) |
| 2035 Market Size | 663.94 (USD Billion) |
| CAGR (2025 - 2035) | 9.08% |