北米: 最も成長が早い医療システム
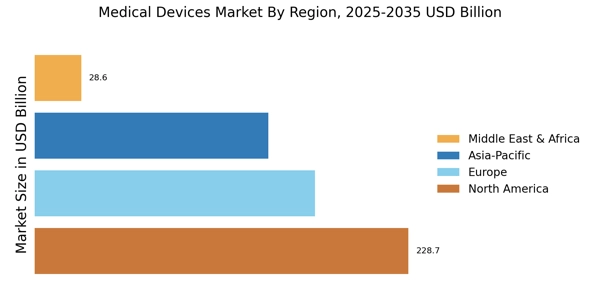
北米は、2024年にグローバル収益の40%以上を占める医療機器市場規模でリードしています。アメリカ合衆国は32%のシェアを持ち、高度な医療インフラと革新的技術の高い採用によって推進されています。一方、カナダは公共医療への強力な投資によって8%を占めています。
- CDCは、米国で年間5000万件以上の外科手術が行われていると報告しており、NIHの資金は年間450億米ドルを超え、WHOは高度な医療技術の広範な採用を強調しており、北米の40%以上の市場シェアを支えています。
アメリカ合衆国は、メドトロニック、アボット、ボストン・サイエンティフィックなどの主要なグローバルベンダーの存在、革新と臨床研究の発展したエコシステムにより、地域市場でのリーダーシップを保持しています。市場は、R&Dへの新たな投資とAIベースの診断ツール、ロボット支援手術、接続された医療機器の迅速な採用により、高い成長率を示しています。
心血管疾患、糖尿病、癌などの慢性疾患の増加は、依然として高度な診断および治療機器の需要を生み出しています。また、在宅医療、遠隔モニタリング、ウェアラブル医療機器への傾向が患者へのサービス提供の方法を変えています。カナダも貢献しており、医療技術の革新と医療のデジタル化に関する好意的な政府政策があります。
米国食品医薬品局(FDA)の規制管理は、製品の品質、安全性、信頼性を保証し、高い消費者信頼を生み出しています。全体として、地域における技術的リーダーシップ、患者の意識、ポジティブな償還構造は、世界の医療機器市場でのさらなる支配の鍵となる要因です。
ヨーロッパ: 新興医療機器
ヨーロッパの医療機器市場規模は2024年に192,569.70百万米ドルと評価され、30%のシェアを持つ第2の地域市場となっています。高齢化する人口、慢性疾患、早期の病気検出と最小限の侵襲的治療手段の高い需要が地域の成長に寄与しています。
- WHOヨーロッパは、60歳以上の人口が25%以上であると報告しており、ECDCはEU諸国で慢性疾患の有病率が増加していることを強調しています。30%の市場シェアと192,569.70百万米ドルの評価を持つヨーロッパは、高度な医療機器に対する強い需要を示しています。
ドイツ、イギリス、フランスは、強力な医療施設と国家医療への広範な政府支出に支えられた重要な貢献者です。
- ドイツは医療技術製造が強力で9%を占めており、イギリスはデジタルヘルスの採用が増加しているため7%を占めています。
EU医療機器規則(MDR)は、市場における透明性と患者の安全性を高めていますが、製造業者に対するコンプライアンスコストを増加させています。ヨーロッパの企業は、画像システム、整形外科インプラント、心臓デバイスの革新においてリードしています。
さらに、すべての医療システムにおけるデジタル変革の傾向は、AIベースの診断技術、ウェアラブル健康ガジェット、eヘルスアプリケーションの使用を促進しています。持続可能性と環境に優しい製造プロセスも地域の議題に上がっており、EUのグリーンディール政策に沿っています。
アジア太平洋: 医療システムの発展
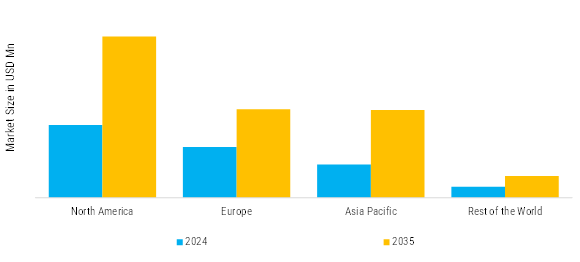
アジア太平洋地域は、医療インフラの成長、都市化、医療意識の高まりにより、医療機器市場が最も急成長しています。主な貢献者には、中国、日本、インド、韓国が含まれ、可処分所得の増加と医療システムの合理化を図る政府政策の恩恵を受けています。地域内の患者数の多さと慢性疾患(糖尿病や心血管疾患)の発生率の増加が、安価な診断および治療機器の需要を高めています。
- 中国は市場価値が57,771百万米ドルで9%をリードしており、急速な医療拡大に支えられています。一方、インドは医療投資の増加と患者人口の成長により5%を占めています。
中国と日本は国際的な製造および革新の中心地であり、国内生産は高く、AIベースの医療技術への大規模な投資があります。一方、インドでは「Make in India」プログラムと医療政策改革の支援を受けて、新興の医療機器製造エコシステムが外国投資の源となっています。手頃な価格とアクセスの向上により、ポータブルおよび在宅医療機器の採用が急増しています。
さらに、オンライン医療スタートアップやテレメディスンポータルの出現が、農村部やサービスが行き届いていない地域での医療提供を改善しています。医療観光も、特にタイ、シンガポール、インドで市場の成長を助けています。アジア太平洋市場は、規制フレームワークが成熟し続け、医療支出が増加する中で、今後数年で世界の医療機器革新と大量生産の主要な源となる可能性があります。
その他の地域: 新興の良好な医療機器
その他の地域(RoW)セグメント - ラテンアメリカ、中東、アフリカ - は、医療機器の成長に良好な地域として浮上しています。市場の成長は、医療インフラの進展、病気に対する意識の向上、病院の近代化への投資の増加によって影響を受けています。主要な市場には、ブラジル、メキシコ、サウジアラビア、UAE、南アフリカが含まれ、現在の医療改革と医療技術の使用促進に関する政府の取り組みがあります。
感染症、慢性疾患、手術の増加率が高く、診断、呼吸、一般外科機器の需要が高まっています。それにもかかわらず、規制システム、均一な償還システムの欠如、輸入機器への依存が一部の地域での成長を妨げています。これらの障壁にもかかわらず、多国籍医療機器企業は、パートナーシップ、現地生産、トレーニングを通じて地域での存在感を高めています。
中東は新しい病院施設や電子医療への積極的な投資を行っており、アフリカとラテンアメリカは国際的な支援と政府ビジネスの取り組みの恩恵を受けています。医療観光の発展と民間セクターの関与の増加は、今後10年間でRoW市場を世界の医療機器産業の最も重要な成長の地平線の1つにする可能性があります。