药物发现中的人工智能市场摘要
p根据 MRFR 分析,2024 年药物发现中的人工智能市场规模估计为 44 亿美元,预计到 2035 年将增长到 250 亿美元,预测期内(2025-2035 年)的复合年增长率 (CAGR) 约为 17.11%。
药物发现中的人工智能市场关键趋势重点介绍
p在几个关键市场驱动因素的推动下,药物发现中的人工智能市场正在经历大幅增长。一个关键因素是对加快药物开发流程的需求不断增长,而传统方法通常难以实现。人工智能技术为预测分析提供了先进的解决方案,提高了药物发现的效率。此外,慢性病发病率的上升和对个性化医疗日益增长的需求,推动了人工智能的应用,以增强药物化合物并精准定位有希望的候选药物。
世界各地的监管机构认识到人工智能在减少药物开发时间和成本方面的潜力,也开始建立支持将其整合到研究流程中的框架。这个市场有很多机会有待探索,特别是在医疗保健需求迅速增长的新兴经济体。公司可以利用人工智能来解决特定的本地健康挑战,并投资与学术机构建立合作伙伴关系,开展创新研究。此外,开放获取的生物医学数据集的日益普及,为人工智能系统提供了一个独特的机会,可以提高其在药物发现中的学习能力和准确性。
最近的趋势表明,制药公司越来越多地在其研究流程中采用机器学习算法和自然语言处理。这种转变正在增强目标识别并优化临床试验设计,从而带来更好的药物性能结果。人工智能技术公司和制药公司之间的合作计划日益普遍,显示出协同增长的趋势。随着技术进步推动全球药物发现和开发流程发生变革,市场对人工智能在药物发现领域的兴趣预计将日益增长。
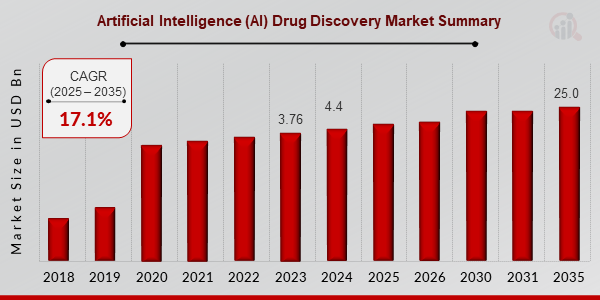
来源:初步研究、二次研究、MRFR 数据库和分析师评论
人工智能药物发现市场驱动因素
h3
个性化医疗需求不断增长 p医疗保健系统内向个性化医疗框架的日益转变推动了人工智能药物发现市场的发展。随着医疗保健提供者越来越认识到根据个体患者基因图谱定制治疗方案的益处,对高级数据分析技术的需求也随之激增。研究表明,精准医疗在针对特定基因标记时可以改善30%或更多患者的治疗效果。
美国国立卫生研究院等机构正在积极推动个性化医疗整合计划,并已投入超过10亿美元支持基因研发项目。这一趋势预计将加速人工智能在药物研发中的应用,因为人工智能算法可以分析海量数据集,确定适合个体患者的合适治疗靶点,从而推动市场增长。
药物研发投资不断增加
p由于制药公司加大对研发的投资,药物研发市场的人工智能正呈现显著上升趋势。根据美国药品研究与制造商协会的数据,仅在2021年,美国公司就在研发方面投资了约830亿美元。
这笔巨额资金投入凸显了人们对创新药物研发途径的日益关注,而人工智能正成为提高药物研发效率的关键技术。辉瑞和强生等大型制药公司正在采用人工智能方法来简化其开发流程,从而减少时间和成本并推动整个市场向前发展。
机器学习和数据分析的进步
p
机器学习和数据分析的重大进步已成为药物发现市场人工智能的关键驱动力。机器学习方法可以解读复杂的生物数据,有助于预测药物相互作用和疗效。随着全球分析市场预计将大幅扩张,药物开发向数据驱动决策的转变预计将进一步扩大。IBM 和 Google 等领先组织正在大力投资人工智能技术,创建复杂的算法,彻底改变药物研究的方式。这种变革性格局极大地增强了识别新型化合物和简化药物开发临床前和临床阶段的能力,推动了市场增长。
药物发现市场中的人工智能细分洞察
h3
药物发现市场中的人工智能应用洞察 p药物发现市场中的人工智能正在经历大幅增长,特别强调应用领域,涵盖目标识别、先导物优化、药物再利用、临床试验和临床前测试等各个关键领域。靶标识别占据着显著的地位,其市场价值在 2024 年为 8.15 亿美元,在 2035 年为 46.12 亿美元,强调了其在药物发现早期阶段的重要性,在该阶段,确定正确的生物靶标对于成功的结果至关重要。
先导化合物优化紧随其后,2024 年的估值为 8.96 亿美元,预计在 2035 年将达到 50.16 亿美元;这一领域至关重要,因为它可以增强现有的候选药物,确保在进行临床试验之前提高疗效并减少副作用。药物再利用的市场价值在 2024 年将达到 9.78 亿美元,到 2035 年将增至 57.44 亿美元,这标志着一种战略方法,即对现有药物进行新治疗用途评估,从而减少传统药物开发通常涉及的时间和成本。
临床试验应用在 2024 年的价值为 14.67 亿美元,预计在 2035 年将达到 80.91 亿美元,在评估药物安全性和有效性方面发挥着至关重要的作用,而人工智能技术有助于更好地选择患者和设计试验,这在日益复杂的临床环境中至关重要。临床前测试的估值似乎较小,2024 年为 2.44 亿美元,预计到 2035 年将增长到 15.37 亿美元,这凸显了在人体试验前进行详尽测试以确保药物安全性和可行性的必要性。
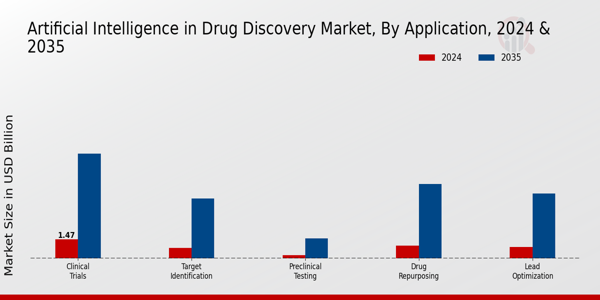
来源:一手资料研究、二手资料研究、MRFR 数据库和分析师评论
人工智能在药物发现市场的技术洞察
p人工智能在药物发现市场按技术划分,涵盖机器学习、自然语言处理深度学习、知识图谱和机器人过程自动化等关键类别。机器学习通过分析大量数据集来识别潜在的候选药物,从而提高药物开发过程的效率和准确性,发挥着关键作用。自然语言处理对于从科学文献中提取有价值的见解至关重要,有助于加快研究进程。
深度学习对预测模型做出了重大贡献,从而可以实现更有针对性的治疗。知识图谱通过可视化不同数据集之间的连接来简化数据关系,从而提高药物发现的决策效率。最后,机器人流程自动化优化了重复性任务,使研究人员能够专注于创新。
人工智能在药物发现市场中的最终用途洞察
p按最终用途划分的全球药物发现人工智能涵盖制药公司、生物技术公司、研究机构和学术机构。制药公司利用人工智能来提高药物开发流程的效率和准确性,从而缩短新疗法上市的时间并降低成本。生物技术公司也发挥着至关重要的作用,他们利用人工智能来简化新型候选药物的发现并优化临床试验,从而显著提高成功率。
研究机构利用人工智能来增强其在药物发现方面的研究能力,为创新和突破创造了巨大的机会。学术机构越来越多地将人工智能融入其教育课程,为下一代科学家提供未来行业发展所需的基本技能。
个性化医疗需求的不断增长以及加速药物开发的必要性,正在推动全球药物发现领域人工智能市场的扩张。技术进步和研发投入的不断增加显著促进了市场增长,凸显了每个终端用户在促进合作转变药物发现实践方面的关键作用。
药物发现市场人工智能工作流程洞察
p药物发现市场人工智能的工作流程涵盖各种组成部分,包括数据挖掘、预测模型、临床数据管理和分析开发。数据挖掘对于从海量数据集中提取有价值的见解至关重要,可帮助研究人员识别潜在的候选药物。
预测模型有助于根据历史数据预测结果,这对于药物开发的明智决策至关重要。临床数据管理可确保临床试验数据的完整性和可访问性,从而促进更顺利的试验和监管审批。分析方法的开发仍然至关重要,因为它直接影响到识别治疗化合物的有效性和准确性。
药物研发中的人工智能市场区域洞察
p北美地区处于领先地位,2024 年该市场的估值为 17.6 亿美元。预计到 2035 年,该市场的估值将达到 101.1 亿美元,这表明其在由先进研究和技术举措驱动的市场动态中占据主导地位。紧随其后的是欧洲,2024 年该市场的估值为 11 亿美元,预计将增长至 63 亿美元,这反映出该地区对医药创新和监管支持的高度重视。
亚太地区 2024 年的价值为 9.2 亿美元,预计将达到 53.2 亿美元,在医疗保健需求不断增长的背景下,亚太地区将成为人工智能技术投资的中心。南美、中东和非洲的市场份额较小,分别为 0.22 亿美元和 0.24 亿美元,但预计到 2035 年将扩大到 13 亿美元和 18.7 亿美元。尽管数字较小,但这些地区正在利用当地合作伙伴关系和投资激励措施来促进人工智能驱动的药物发现领域的增长。
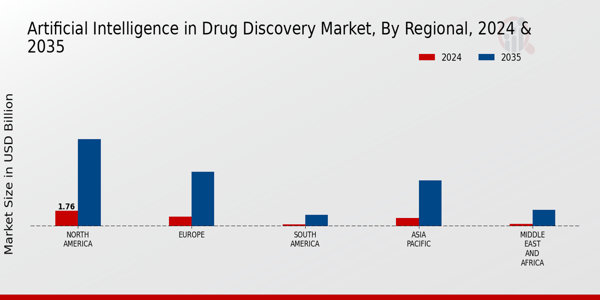
来源:初步研究、二次研究、MRFR 数据库和分析师评论
人工智能药物发现市场主要参与者和竞争洞察:
p全球药物开发人工智能市场已经成为知名制药和技术企业的首要任务,这些企业旨在优化其药物发现流程并缩短上市时间。该领域利用先进的机器学习算法和数据分析,显著提高了识别潜在候选药物的效率。随着定制药物和新药需求的不断增长,该领域的竞争也愈演愈烈,战略合作、技术改进和知识产权开发尤为突出。
目前,各公司正在探索将人工智能应用于药物识别、临床试验和上市后监测的优化。该领域在研发方面投入了大量资金,专注于开发能够预测药物相互作用和副作用的先进人工智能平台,旨在优化药物开发过程的每个阶段。
诺华公司凭借其对创新和技术应用的承诺,在药物发现市场的人工智能领域脱颖而出。凭借强大的研发渠道,诺华公司已将人工智能融入其药物发现活动的各个方面,从而实现更高效的筛选和优化分子药物设计。与科技公司和学术机构的战略合作巩固了其市场地位,使诺华能够利用最先进的人工智能解决方案。
诺华的优势在于其涵盖各种治疗领域的广泛产品组合,以及其利用人工智能重新利用现有药物的能力,从而有可能加快药物发现过程。这种前瞻性的方法加上诺华已建立的行业声誉,巩固了其在利用人工智能技术进行药物发现全球应用方面的竞争优势。
Atomwise 是药物发现市场中人工智能的另一个关键参与者,因其在药物设计和开发中对人工智能的创新应用而闻名。该公司的专有技术利用深度学习算法来预测潜在药物分子的有效性,大大加快了药物发现的初始阶段。 Atomwise 在与全球众多制药公司和研究机构建立战略合作伙伴关系方面取得了显著进展,使其技术广泛应用于各个治疗领域。
公司的优势在于其独特的人工智能平台,该平台提供高效的虚拟筛选服务,在候选药物的识别方面取得了很高的成功率。Atomwise 还开展了多项引人注目的合作与并购,巩固了其市场地位,增强了其产品组合和药物研发能力。专注于人工智能和战略扩张,使 Atomwise 在快速发展的药物发现领域保持竞争地位,在全球范围内推动创新和效率。
药物发现人工智能市场的主要公司包括:
ul
诺华 Atomwise Recursion Pharmaceuticals 辉瑞 微软 Insilico Medicine IBM Biogen 阿斯利康 谷歌 DeepMind Exscientia 百时美施贵宝 h2
药物发现人工智能市场发展 p全球药物发现人工智能市场正在经历显著增长,领先的制药公司包括诺华、辉瑞和阿斯利康利用人工智能提升药物开发效率。Atomwise 继续保持领先地位,其 AtomNet® 平台可对数十亿种化合物进行虚拟筛选,从而显著提升早期药物识别速度。Insilico Medicine 和 DeepMind(通过 Isomorphic Labs)等其他公司已获得大量融资,彰显了该行业的创新能力和日益增长的投资者信任。Exscientia 与百时美施贵宝于 2021 年 5 月宣布合作,这是双方合作的重要里程碑。该协议价值超过 12 亿美元,致力于利用人工智能提升各种治疗靶点的药物开发效率。此次合作已成功完成PKC-θ抑制剂 (EXS4318) 的首次人体试验,凸显了AI驱动创新在临床开发中带来的切实益处。
尽管市场普遍乐观,但必须澄清的是,与某些报道相反,IBM 在 2023 年 8 月并未收购任何 AI 药物研发公司。2022 年,IBM 重组了其早期的医疗保健 AI 项目,并最终成立了 Merative。总体而言,在战略合作伙伴关系、不断发展的平台以及对技术集成的高度重视的推动下,药物研发领域的人工智能技术将持续增长。
药物研发市场细分洞察中的人工智能
h3
药物研发市场应用展望中的人工智能 ul
靶点识别 先导化合物优化 药物再利用 临床试验 临床前测试 h3
药物研发市场技术展望中的人工智能 ul
机器学习 自然语言处理 深度学习 知识图谱 机器人流程自动化 h3
人工智能在药物研发市场中的最终用途展望 ul
制药公司 生物技术公司 研究机构 学术机构 h3
人工智能在药物研发市场中的工作流程展望 ul
数据挖掘 预测模型 临床数据管理 分析开发 h3
人工智能在药物研发市场中的区域展望 ul
北美 欧洲 南美 亚太地区 中东和非洲













