Medical Device Regulatory Affairs Outsourcing Market
ID: MRFR/MED/37232-HCR
100 Pages
Nidhi Mandole
Last Updated: April 06, 2026
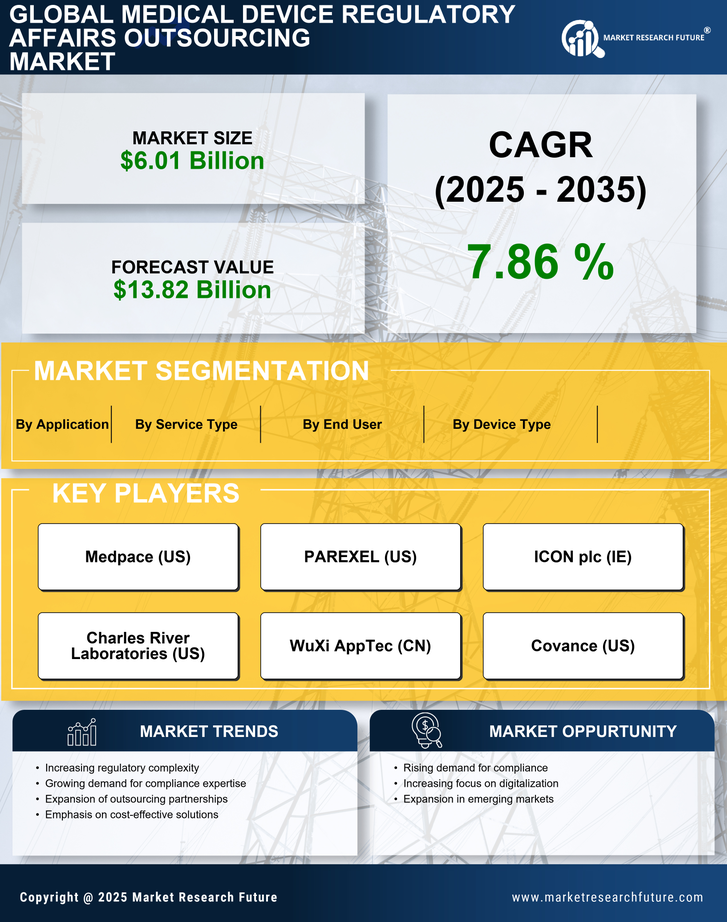
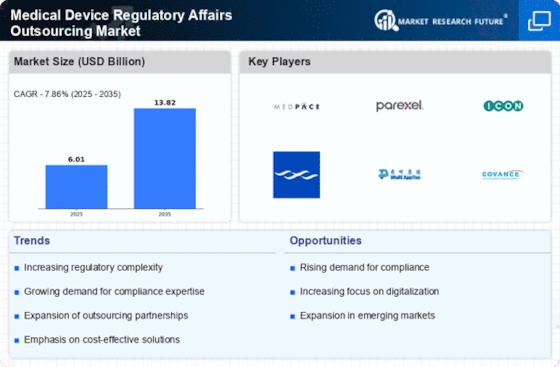
Medical Device Regulatory Affairs Outsourcing Market Research Report By Service Type (Regulatory Consulting, Regulatory Submission, Labeling and Advertising Compliance, Clinical Trial Application, Quality Management System), By Device Type (In Vitro Diagnostics, Cardiovascular Devices, Orthopedic Devices, Neurological Devices, Surgical Instruments), By End User (Medical Device Manufacturers, Pharmaceutical Companies, Research Organizations, Contract Research Organizations, Academic Institutions), By Application (Diagnostics, Therapeutics, Monitoring, Surgical Procedures, Patient Management) and By Regional (North America, Europe, South America, Asia Pacific, Middle East and Africa) - Growth & Industry Forecast 2025 To 2035