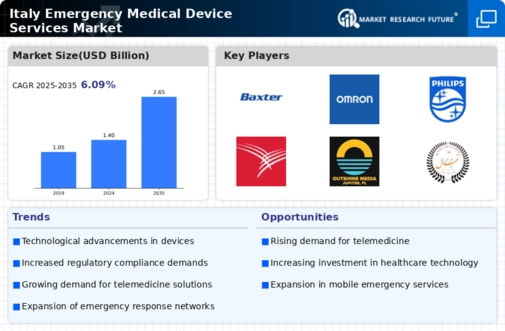
Italy Healthcare Market Segmentation
Healthcare By Application (USD Billion, 2026-2035)
- Cardiac Monitoring
- Respiratory Support
- Trauma Care
- Patient Transport
- Defibrillation
Healthcare By End Use (USD Billion, 2026-2035)
- Hospitals
- Ambulance Services
- Emergency Medical Services
- Home Care
- Urgent Care Centers
Healthcare By Device Type (USD Billion, 2026-2035)
- Automated External Defibrillators
- Ventilators
- Monitors
- Stretchers
- First Aid Kits
Healthcare By Technology (USD Billion, 2026-2035)
- Wireless Technology
- Telemedicine
- Portable Devices
- Wearable Devices
- Mobile Applications
Healthcare By Regulatory Compliance (USD Billion, 2026-2035)
- ISO Standards
- CE Marking
- FDA Approval
- Local Health Regulations
- Safety Certifications