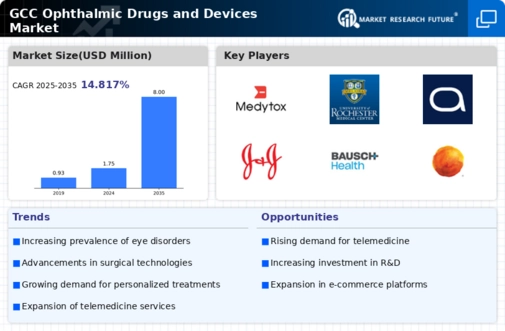
GCC Healthcare Market Segmentation
Healthcare By Application (USD Billion, 2026-2035)
- Glaucoma Treatment
- Anti-Inflammatory
- Infection Control
- Dry Eye Treatment
- Surgical Procedures
Healthcare By Device Type (USD Billion, 2026-2035)
- Intraocular Lens
- Surgical Instruments
- Diagnostic Equipment
- Contact Lenses
- Ocular Drug Delivery Systems
Healthcare By Formulation Type (USD Billion, 2026-2035)
- Topical Solutions
- Injectable Solutions
- Ocular Implants
- Sustained Release Formulations
- Ophthalmic Gels
Healthcare By End User (USD Billion, 2026-2035)
- Hospitals
- Clinics
- Home Care Settings
- Research Institutions
- Pharmacies