Bioanalytical Testing Services Market Summary
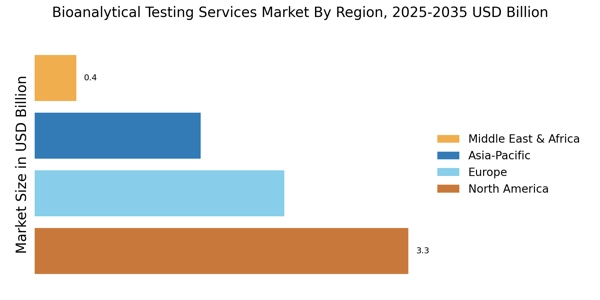
As per Market Research Future analysis, the Bioanalytical Testing Services Market was estimated at 4.6 USD Billion in 2025. The Bioanalytical Testing Services industry is projected to grow from 5.0 USD Billion in 2026 to 10.9 USD Billion by 2035, exhibiting a compound annual growth rate (CAGR) of 9.01% during the forecast period 2026 - 2035. North America led the market with over 71.74% share, generating around USD 3.3 billion in revenue.
The Bioanalytical Testing Services Market is primarily driven by increasing complexity in drug development, rising demand for accurate pharmacokinetic and biomarker analysis, and growing outsourcing by pharmaceutical companies to ensure regulatory compliance, improve efficiency, and accelerate clinical trial timelines globally.
According to the World Health Organization (WHO), over 2 million new cancer cases annually in Europe alone and rising global disease burden are accelerating drug development needs, while the Institute for Health Metrics and Evaluation (IHME) highlights increasing clinical research activities, boosting demand for advanced bioanalytical testing services.
Key Market Trends & Highlights
The Bioanalytical Testing Services Market is experiencing robust growth driven by technological advancements and increasing regulatory compliance.
- North America accounted for over 71.74% revenue share in 2024, driven by strong clinical research infrastructure.
- Mass spectrometry holds 41% share, reflecting high adoption for precise quantitative bioanalytical testing globally.
- ADME testing leads with 38% share, driven by increasing demand for drug metabolism and pharmacokinetic analysis.
- Pharmaceuticals dominate applications with 62% share, supported by expanding global drug development pipelines and regulatory requirements.
Market Size & Forecast
| 2025 Market Size | 4.6 (USD Billion) |
| 2035 Market Size | 10.9 (USD Billion) |
| CAGR (2025 - 2035) | 9.01% |
Major Players
Companies such as LabCorp (US), Charles River Laboratories (US), Eurofins Scientific (LU), PPD (US), Covance (US), Syneos Health (US), Quotient Limited (GB), Medpace (US), Wuxi AppTec (CN) are some of the major participants in the global market.