バイオアナリティカルテストサービス市場 概要
MRFRの分析によると、バイオアナリティカルテストサービス市場は2024年に73.52億米ドルと推定されています。バイオアナリティカルテストサービス業界は、2025年に78.79億米ドルから2035年までに157.5億米ドルに成長する見込みで、2025年から2035年の予測期間中に年平均成長率(CAGR)は7.17を示します。
主要な市場動向とハイライト
バイオアナリティカルテストサービス市場は、技術の進歩と規制遵守の増加により、堅調な成長を遂げています。
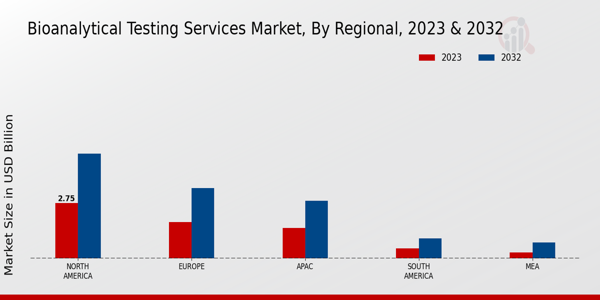
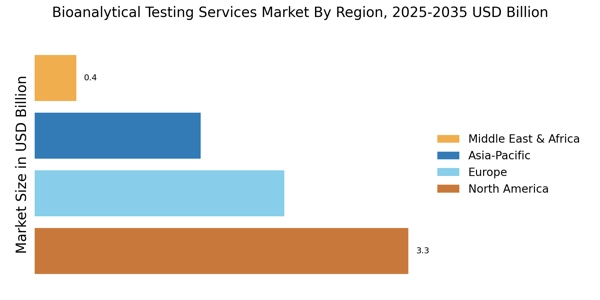
- 北米は、強力なバイオ医薬品セクターに支えられ、バイオアナリティカルテストサービスの最大の市場であり続けています。アジア太平洋地域は、医療および臨床研究への投資の増加により、最も成長している地域として浮上しています。ADMEテストは市場を支配し続けており、バイオマーカー検査は重要な成長分野として急速に注目を集めています。バイオ医薬品の需要の高まりと分析技術の進歩は、市場の拡大の重要な推進要因です。
市場規模と予測
| 2024 Market Size | 7.352 (米ドル十億) |
| 2035 Market Size | 157.5億ドル |
| CAGR (2025 - 2035) | 7.17% |
主要なプレーヤー
ラボコープ(米国)、チャールズリバーラボラトリーズ(米国)、ユーロフィンズサイエンティフィック(ルクセンブルク)、PPD(米国)、コバンス(米国)、シネオスヘルス(米国)、クオティエントリミテッド(英国)、メドペース(米国)、ウーシーアプテック(中国)