US Medical Device Market Segmentation
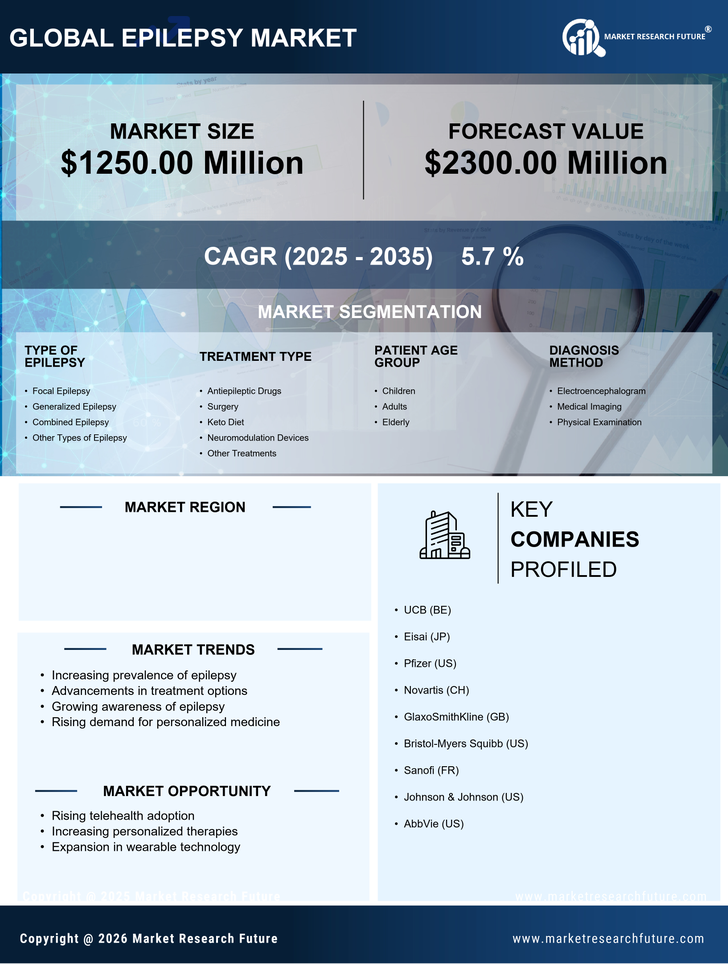
Medical Device By Type of Epilepsy (USD Million, 2025-2035)
- Focal Epilepsy
- Generalized Epilepsy
- Combined Epilepsy
- Other Types of Epilepsy
Medical Device By Treatment Type (USD Million, 2025-2035)
- Antiepileptic Drugs
- Surgery
- Keto Diet
- Neuromodulation Devices
- Other Treatments
Medical Device By Patient Age Group (USD Million, 2025-2035)
- Children
- Adults
- Elderly
Medical Device By Diagnosis Method (USD Million, 2025-2035)
- Electroencephalogram
- Medical Imaging
- Physical Examination