Epilepsy Market Summary
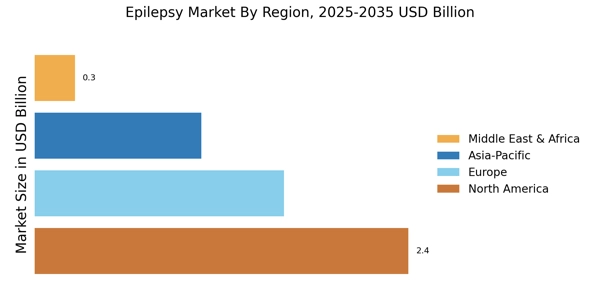
As per Market Research Future analysis, the Epilepsy Market Size was estimated at 5.33 USD Billion in 2024. The Epilepsy industry is projected to grow from 5.617 USD Billion in 2025 to 9.496 USD Billion by 2035, exhibiting a compound annual growth rate (CAGR) of 5.3% during the forecast period 2025 - 2035. North America led the market with over 43.15% share, generating around USD 2.3 billion in revenue.
The Epilepsy Market is primarily driven by increasing global prevalence of neurological disorders and improved diagnosis rates, alongside growing demand for advanced therapies and personalized treatment approaches that enhance seizure control, patient outcomes, and long-term disease management across healthcare systems.
According to the World Health Organization (WHO), epilepsy affects around 50 million people globally, while the Institute for Health Metrics and Evaluation (IHME) highlights a rising neurological disease burden, reinforcing demand for improved treatment access and innovative epilepsy therapies worldwide.
Key Market Trends & Highlights
The Epilepsy Market is experiencing a dynamic evolution driven by technological advancements and a focus on personalized care.
- Focal epilepsy leads with 64% share, reflecting higher prevalence and strong pipeline of targeted therapies.
- North America holds 43.15% share, while Europe accounts for 28.14% share valued at USD 1.5 billion in 2024.
- Epilepsy affects 50 million people globally, significantly driving demand for advanced neurological treatments and diagnostic solutions.
- Antiepileptic drugs dominate with 68% share, supported by widespread clinical use and established therapeutic effectiveness.
Market Size & Forecast
| 2024 Market Size | 5.33 (USD Billion) |
| 2035 Market Size | 9.496 (USD Billion) |
| CAGR (2025 - 2035) | 5.39% |
Major Players
Companies such as UCB (BE), Eisai (JP), Novartis (CH), Pfizer (US), GlaxoSmithKline (GB), AbbVie (US), Sanofi (FR), Lundbeck (DK), Teva (IL) are some of the major participants in the global market.