North America : Innovation and Market Leadership
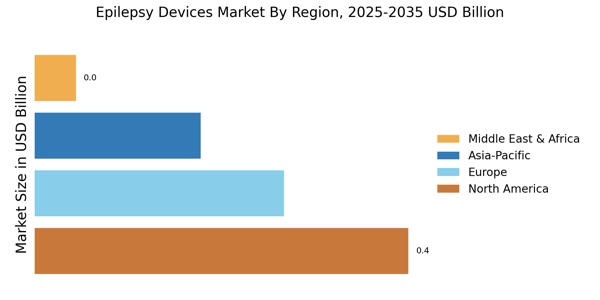
North America is the largest market for epilepsy devices, holding approximately 45% of the global share. The region benefits from advanced healthcare infrastructure, high prevalence of epilepsy, and significant investments in research and development. Regulatory support from agencies like the FDA accelerates the approval of innovative devices, driving market growth. The increasing awareness of epilepsy management and technological advancements further fuel demand.
The United States leads the North American market, with key players such as Medtronic, Abbott, and Boston Scientific dominating the landscape. These companies are continuously innovating, focusing on developing advanced neurostimulation devices and monitoring systems. The competitive environment is characterized by strategic partnerships and collaborations aimed at enhancing product offerings and expanding market reach, ensuring a robust presence in the epilepsy devices sector.
- CDC data shows epilepsy affects approximately 3.4 million people in the United States, including 3 million adults and 470,000 children. Soterix Medical clinical neurostimulation programs report up to 40% improvement in seizure control outcomes, supporting strong adoption of advanced epilepsy devices across U.S. healthcare systems.
Europe : Emerging Market with Growth Potential
Europe is the second-largest market for epilepsy devices, accounting for approximately 30% of the global share. The region is witnessing a surge in demand due to increasing awareness of epilepsy and advancements in medical technology. Regulatory bodies, such as the European Medicines Agency (EMA), are actively promoting innovative solutions, which is expected to enhance market growth. The rising incidence of epilepsy and supportive healthcare policies are key drivers in this region.
Leading countries in Europe include Germany, France, and the UK, where significant investments in healthcare infrastructure are observed. The competitive landscape features major players like LivaNova and NeuroPace, who are focusing on developing cutting-edge devices. The presence of a well-established healthcare system and a growing emphasis on patient-centric solutions are further propelling the market forward, making Europe a vital region for epilepsy device manufacturers.
- EMA-supported clinical evaluations highlight increasing approvals of neurostimulation devices, with treatment-resistant epilepsy affecting nearly 6 million people across Europe. Regulatory advancements have reduced approval timelines by up to 25%, enabling faster access to innovative epilepsy monitoring and neurostimulation technologies across key European healthcare markets.
Asia-Pacific : Rapidly Growing Market Segment
Asia-Pacific is an emerging powerhouse in the market, holding around 20% of the global share. The region is characterized by a growing population, increasing healthcare expenditure, and rising awareness of neurological disorders. Countries like China and India are witnessing a surge in demand for advanced medical devices, driven by the need for better healthcare solutions. Government initiatives aimed at improving healthcare access are also contributing to market growth.
China and Japan are the leading countries in this region, with a strong presence of local and international players. Companies like ElectroCore and Soterix Medical are expanding their operations to cater to the growing demand. The competitive landscape is evolving, with a focus on innovation and affordability, ensuring that epilepsy devices become more accessible to patients across the region. The Asia-Pacific market is poised for significant growth in the coming years, driven by technological advancements and increasing healthcare investments.
Middle East and Africa : Untapped Market with Potential
The Middle East and Africa represent an untapped market for epilepsy devices, holding approximately 5% of the global share. The region faces challenges such as limited healthcare infrastructure and awareness, but there is a growing recognition of the need for advanced medical solutions. Increasing investments in healthcare and government initiatives aimed at improving access to treatment are expected to drive market growth in the coming years.
Countries like South Africa and the UAE are leading the way in adopting new medical technologies. The competitive landscape is gradually evolving, with both local and international players looking to establish a foothold. Companies are focusing on education and awareness campaigns to promote epilepsy management solutions, which is crucial for expanding the market. The potential for growth in this region is significant, as healthcare systems continue to develop and improve.