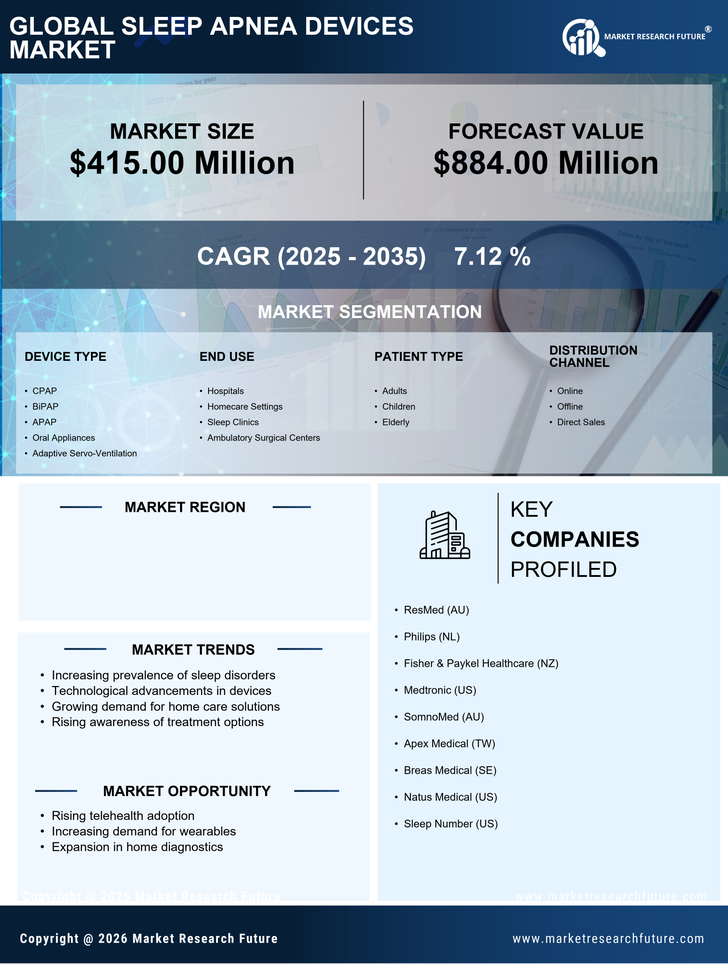
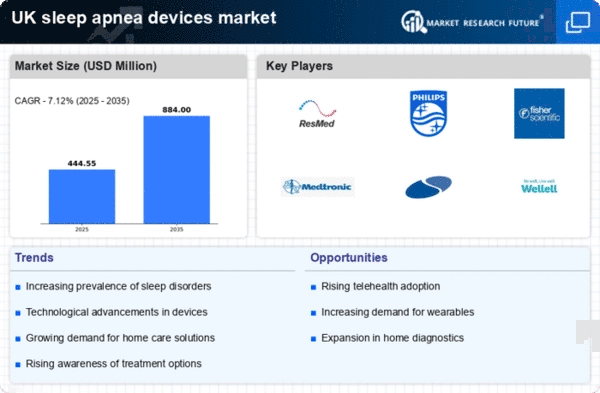
UK Medical Device Market Segmentation
Medical Device By Device Type (USD Million, 2025-2035)
- CPAP
- BiPAP
- APAP
- Oral Appliances
- Adaptive Servo-Ventilation
Medical Device By End Use (USD Million, 2025-2035)
- Hospitals
- Homecare Settings
- Sleep Clinics
- Ambulatory Surgical Centers
Medical Device By Patient Type (USD Million, 2025-2035)
- Adults
- Children
- Elderly
Medical Device By Distribution Channel (USD Million, 2025-2035)
- Online
- Offline
- Direct Sales