South Korea Ophthalmic Drugs Market
ID: MRFR/Pharma/43557-HCR
200 Pages
Vikita Thakur
Last Updated: April 06, 2026
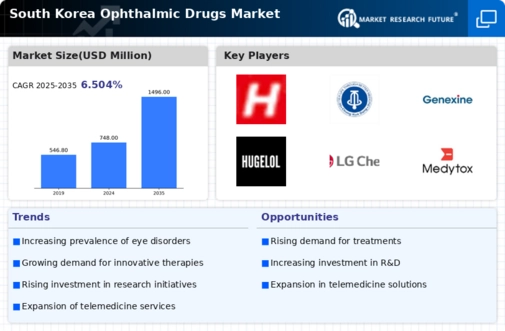
South Korea Ophthalmic Drugs Market Research Report By Dosage Form (Eye Drops, Eye Ointments, Eye Solutions, Gels, Capsules Tablets, Others), By Product Type (Prescription Drug, OTC Drug), By Drug Class (Antibiotics, Anti-Inflammatory, Anti-Glaucoma, Others), By Indication (Glaucoma, Retinal Disorder, Dry Eye, Eye Allergy, Others) and By Distribution Channels (Hospital pharmacies, Drug stores, Online pharmacies, Others) -Forecast to 2035