South America Life Science Analytics Market
ID: MRFR/LS/49375-HCR
200 Pages
Vikita Thakur
Last Updated: February 06, 2026
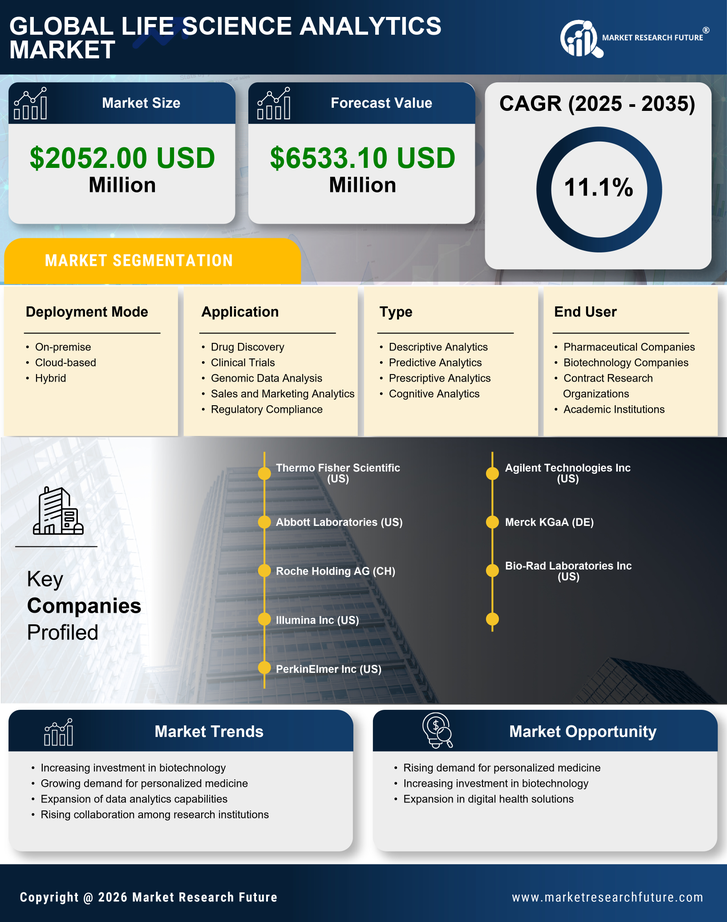
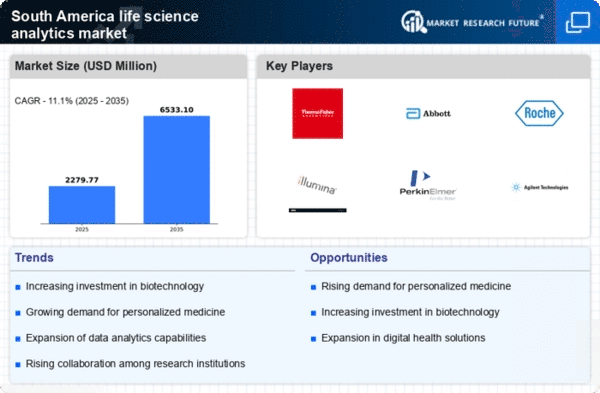
South America Life Science Analytics Market Research Report: Size, Share, Trend Analysis By Types of Analytics (Predictive Analytics, Descriptive Analytics, Prescriptive Analytics), By Applications (Research & Development, Pharmacovigilance, Regulatory Compliance, Sales & Marketing, Supply Chain Optimization), By Component (Software, Services, Hardware), By Deployment Model (On-Premise, Cloud-Based), By End Users (Hospitals, Clinics, Others) and By Regional (Brazil, Mexico, Argentina, Rest of South America) - Growth Outlook & Industry Forecast 2025 To 2035