Market Analysis
In-depth Analysis of Somatostatin Analogs Market Industry Landscape
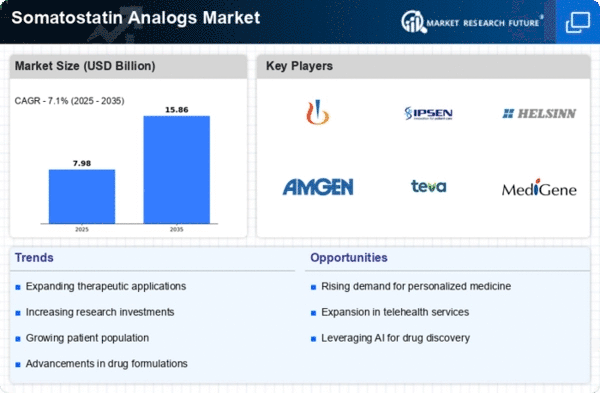
The dynamics and growth trajectory of the Somatostatin Analogs Market are shaped by different factors that are collectively taken together. One of its main drivers is the prevalence of conditions such as acromegaly, neuroendocrine tumors as well as Cushing's disease, for which they are used as therapeutic agents. The market for somatostatin analogs is influenced by advances made in medical research and technology. Ongoing studies seek to unravel molecular pathways implicated in endocrine diseases; hence, better design of somatostatin with greater effectiveness is needed. The resulting new analogs combined with advances in drug delivery systems contribute to the expansion of the market, providing health professionals with improved means of managing and treating these diseases. The development and commercialization of somatostatin analogs are deeply influenced by regulatory considerations as well. Stringent regulatory standards assure healthcare providers and patients of the safety and efficacy of these treatments. Economic factors significantly impact the somatostatin analogs market, which determines the cost of production and pricing strategies used by companies in this industry. Higher economic growth rates coupled with political stability tend to heighten healthcare spending, thereby enhancing access to such treatments. On the other hand, economic recessions might have a bearing on healthcare spending decisions, thus affecting market trends. In certain areas, particularly where economic factors play a role in healthcare decisions, costs and availability of somatostatin analogs become important factors to consider. The competitive landscape among pharmaceutical companies is another critical market factor. This intense competition stimulates innovation as firms try to develop more advanced somatostatin analogs for a competitive edge. Consumer preferences, as well as changing trends in healthcare across different markets, also influence sales within the field of Somatostatin Analogues. Patient preferences for convenience, lower side effect profiles, and specific therapies drive product development projects. Furthermore, policies related to the overall structure of healthcare systems, together with reimbursement, have a great role in shaping the adoption of the Somatostatin Analogs Market. Both accessibilities to these products and reimbursement policies are determinants for both healthcare providers'/consumers' choices, hence influencing adoption or non-adoption or specific ones concerning the somatostatin analog.


















Leave a Comment