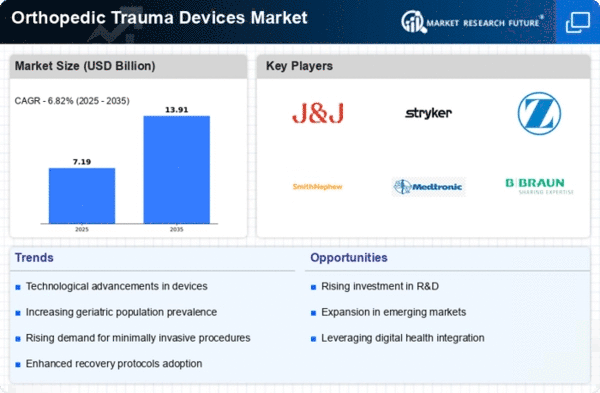
Market Analysis
In-depth Analysis of Orthopedic Trauma Devices Market Industry Landscape
The dynamics within the orthopedic trauma devices industry mirror a complex interplay between various factors which determine its growth and development. As of the latest analysis, this sector has been expanding at a consistent rate because there is an increasing number of orthopedic injuries and fractures caused by issues like aging population and increased involvement in sporting activities. Moreover, there are high instances of road accidents leading to a high demand for orthopedic trauma devices since they play critical role during treatment and rehabilitation processes involving patients living with musculoskeletal conditions.
Furthermore, the market is highly influenced by awareness among older people on physical activity maintenance. To this effect, people struggling to maintain their mobility and quality of life are increasingly seeking orthopedic trauma devices which can provide effective solutions for a long time. In developed areas where healthcare infrastructure and disposable income levels are high, there is an increased adoption of advanced orthopedic treatments. On the other hand, the market dynamics face challenges because of expensive orthopedic trauma devices. Despite their advantages, these devices still remain unaffordable in emerging economies. Consequently, many individuals globally cannot have access to advanced orthopaedic treatments thus affecting market penetration and growth potential in some areas due to this economic barrier. Moreover, the regulatory framework determines how the market moves. For one thing, strict regulation concerning approval of orthopedic trauma devices could have an impact on product development timelines as well as increase overall cost towards introduction of new technologies into the market. These regulations must be effectively managed by manufacturers for them to comply with and enter the market successfully. Geographically speaking; North America and Europe lead in terms of orthopedic trauma devices with developed healthcare infrastructure, significant healthcare spending and established regulatory framework meanwhile Asia-Pacific is becoming an important player supported by increasing health care awareness increasing disposable income level growing aging population that’s why organization focus on expanding geographical coverage in these regions to capitalize on untapped growth potential.



















Leave a Comment