The secondary research process involved comprehensive analysis of regulatory databases, peer-reviewed orthopedic journals, clinical publications, trauma surgery literature, and authoritative healthcare organizations. Key sources included:
Regulatory & Government Bodies:
US Food & Drug Administration (FDA) - Center for Devices and Radiological Health (CDRH)
European Medicines Agency (EMA) - Medical Device Regulation (MDR) databases
Health Canada - Medical Devices Active Licence Listing (MDALL)
Therapeutic Goods Administration (TGA) - Australian Register of Therapeutic Goods (ARTG)
Pharmaceuticals and Medical Devices Agency (PMDA) - Japan medical device approvals
China National Medical Products Administration (NMPA)
UK Medicines and Healthcare products Regulatory Agency (MHRA)
Medical & Professional Associations:
Orthopaedic Trauma Association (OTA)
American Academy of Orthopaedic Surgeons (AAOS)
AO Foundation (Arbeitsgemeinschaft für Osteosynthesefragen)
International Society of Orthopaedic Surgery and Traumatology (SICOT)
American Orthopaedic Association (AOA)
British Orthopaedic Association (BOA)
European Federation of National Associations of Orthopaedics and Traumatology (EFORT)
Healthcare Statistics & Research Organizations:
National Institutes of Health (NIH) - National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS)
National Center for Biotechnology Information (NCBI/PubMed)
Agency for Healthcare Research and Quality (AHRQ) - Healthcare Cost and Utilization Project (HCUP)
Centers for Disease Control and Prevention (CDC) - National Center for Injury Prevention and Control (NCIPC)
World Health Organization (WHO) - Global Health Observatory & Injury/Violence Prevention Data
Organisation for Economic Co-operation and Development (OECD) - Health Statistics
Eurostat - Healthcare Statistics & Hospital Discharge Data
National Trauma Data Bank (NTDB) / American College of Surgeons Trauma Quality Programs
US Department of Transportation - National Highway Traffic Safety Administration (NHTSA) Crash Statistics
Bureau of Labor Statistics (BLS) - Occupational Injuries and Illnesses Data
These sources were utilized to collect trauma incidence statistics, fracture classification data (AO/OTA classification), surgical procedure volumes, regulatory approval pathways (510(k), PMA, CE marking), clinical outcomes studies, trauma center infrastructure data, demographic trends in musculoskeletal injuries, and competitive landscape analysis for internal fixation devices (plates & screws, intramedullary nails, cannulated screws), external fixation devices, and craniomaxillofacial fixation systems.

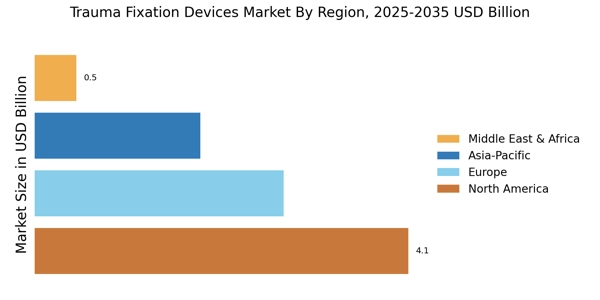
















.png?v=1775475622)