Market Analysis
In-depth Analysis of Ophthalmic Drugs Devices Market Industry Landscape
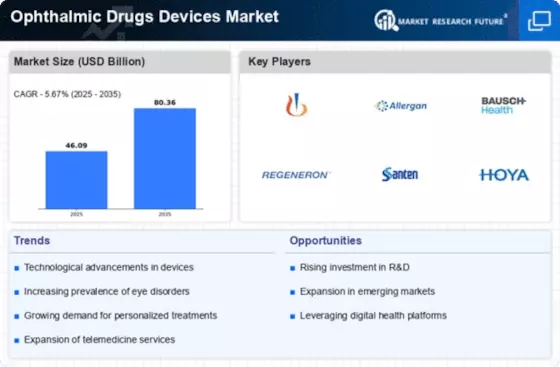
| The market dynamics of the ophthalmic drugs and devices industry are shaped by a complex interplay of various factors, driving growth and influencing trends. One of the primary drivers is the increasing prevalence of eye disorders and diseases globally. With the aging population on the rise, conditions like cataracts, glaucoma, and macular degeneration are becoming more prevalent, propelling the demand for ophthalmic drugs and devices. Moreover, lifestyle changes and the growing incidence of diabetic retinopathy contribute to the expanding market. Technological advancements play a crucial role in shaping the market landscape. Innovations in drug delivery systems, such as sustained-release implants and novel formulations, enhance treatment efficacy and patient compliance. Similarly, cutting-edge devices, including advanced diagnostic tools and surgical equipment, drive the market forward by offering more precise and less invasive treatment options. The adoption of artificial intelligence and machine learning in diagnostics further revolutionizes ophthalmic care, providing faster and more accurate assessments of eye conditions. Global collaborations and strategic partnerships among pharmaceutical companies, medical device manufacturers, and research institutions are fostering a collaborative environment that accelerates research and development activities. This collaborative approach aids in the discovery of novel therapies and the introduction of innovative devices, ultimately expanding the market. Regulatory bodies also play a pivotal role, ensuring the safety and efficacy of ophthalmic drugs and devices through rigorous approval processes, thus maintaining quality standards and fostering market growth. Market dynamics are also influenced by economic factors, with the affordability and accessibility of ophthalmic drugs and devices impacting their adoption rates. Governments and healthcare organizations worldwide are increasingly recognizing the importance of eye health, leading to initiatives and policies that support the development and distribution of ophthalmic solutions. Furthermore, the expansion of healthcare infrastructure in emerging economies contributes to the market's growth, as more people gain access to eye care facilities and treatments. Competitive forces within the industry propel companies to invest in research and development, fostering a continuous cycle of innovation. The race to introduce new and improved products drives market competition, pushing companies to differentiate themselves through technological advancements, pricing strategies, and market expansion initiatives. As a result, consumers benefit from a wider range of treatment options and advanced devices. Challenges also characterize the market dynamics of the ophthalmic drugs and devices sector. Stringent regulatory requirements and the lengthy approval process for new drugs and devices can hinder the speed of innovation and market entry. Additionally, the high development costs associated with bringing new products to market pose a challenge for smaller companies. Economic uncertainties and fluctuations in healthcare budgets further impact market dynamics, influencing investment decisions and market growth trajectories. |



















Leave a Comment