New-Born Screening Market Summary
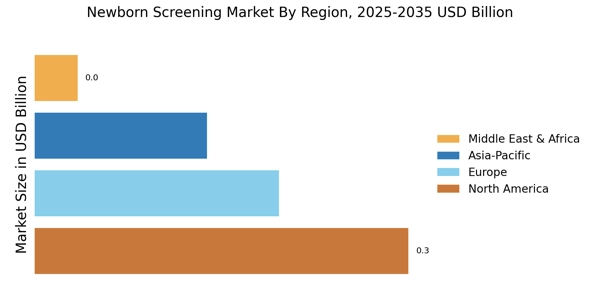
According to Market Research Future analysis, the Newborn Screening Market size was valued at USD 0.58 Billion in 2024. The market is projected to grow from USD 0.6218 Billion in 2025 to USD 1.248 Billion by 2035, registering a CAGR of 7.2% during the forecast period 2025–2035. North America led the New-Born Screening Market with over 53% share, generating around USD 0.3 Billion in revenue.
The Newborn Screening Market is expanding due to rising prevalence of congenital disorders and increasing emphasis on early disease detection. Key trends include advancements in genetic screening technologies, government-led universal screening programs, and growing awareness among parents, enabling timely interventions that improve infant health outcomes and reduce long-term healthcare burden globally.
According to the World Health Organization, newborn screening enables early detection of serious conditions, helping prevent disability and premature death through timely intervention.
Key Market Trends & Highlights
The Newborn Screening Market is poised for substantial growth driven by technological advancements and increasing awareness.
- Technological advancements in screening methods are enhancing the accuracy and efficiency of newborn screening processes.
- North America remains the largest market, while the Asia-Pacific region is emerging as the fastest-growing area for newborn screening.
- Disorders screening continues to dominate the market, whereas congenital heart disease screening is witnessing rapid growth.
- Key market drivers include the rising incidence of congenital disorders and government initiatives supporting expanded screening programs.
Market Size & Forecast
| 2024 Market Size | 0.58 (USD Billion) |
| 2035 Market Size | 1.248 (USD Billion) |
| CAGR (2025 - 2035) | 7.21% |
Major Players
Companies such as PerkinElmer (US), Thermo Fisher Scientific (US), Abbott Laboratories (US), Roche Diagnostics (CH), Agilent Technologies (US), Bio-Rad Laboratories (US), Newborn Screening Laboratory (US), Natus Medical Incorporated (US), Mayo Clinic Laboratories (US) are some of the major participants in the global market.