Market Analysis
In-depth Analysis of New-Born Screening Market Industry Landscape
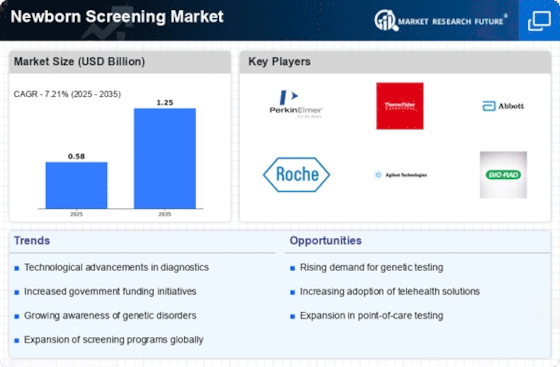
The growth of the newborn screening industry is driven by diverse factors that interact together, making its dynamics very complex. For example, Newborn Screening is an essential component of the healthcare system aimed at detecting life-threatening diseases at the initial stages in neonates. One key driver behind its growth is increasing awareness among parents and medical practitioners about the advantages resulting after having an early diagnosis, with intervention initiated at the earliest moment possible before complications set in. Major advances in screening methods have had a substantial effect on market dynamics. This has increased the accuracy and efficiency of newborn screening by constantly developing new technologies like tandem mass spectrometry, DNA-based testing, and next-generation sequencing. As a result, the number of disorders that may be detected has been expanded while simplifying the process to make it more affordable. Automation and data management solutions have also been integrated, which have stimulated the growth of the newborn screening market by making workflow faster and reducing turnaround time. Government initiatives and policies shape the newborn screening market. In many nations, there are mandatory programs for such screenings that increase the number of newborns tested. It is common for industry players to collaborate or partner with each other, thereby fostering market growth through shared expertise, resources, and technological advancements. This has led to the creation of comprehensive screening solutions that address a broader spectrum of diseases. Furthermore, research institutes form strategic alliances with stakeholders in this sector, hence enhancing knowledge concerning genetic and metabolic problems, leading to further development within newborn screening innovations. The other factor that greatly influences market dynamics is the economic landscape. Thus, the availability and price of newborn screening services are directly influenced by economic development and an increase in healthcare spending. With economic growth, there is more capacity for investment in advanced screening technologies and infrastructure, hence bolstering market expansion. Besides, rising incomes among individuals have enabled many more families to undertake newborn screening, hence increasing market size further. Challenges, however, exist within the newborn screening market. Ethical considerations such as privacy issues and false positives need to be addressed so as to screen babies effectively.



















Leave a Comment