Italy Medical Device Market
ID: MRFR/MED/42601-HCR
200 Pages
Rahul Gotadki
Last Updated: April 06, 2026
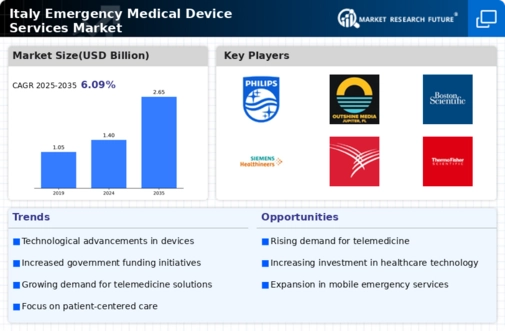
Italy Medical Device Market Research Report By Device Type Outlook (Drug Delivery System, Diagnostic Devices, Invasive Devices, Respiratory Therapeutic Devices, Electronic Medical Device, Others), By Application Outlook (Cardiovascular Diseases, Diagnostic Imaging, Orthopedics, Oncology, Ophthalmology, General Surgery, Respiratory, Dental, Ear, Nose, and Throat (ENT), Neurology, Nephrology Urology, Others) and By End User Outlook (Hospitals Clinics, Ambulatory Surgical Centers, Homecare Settings, Others) - Growth & Industry Forecast 2025 To 2035