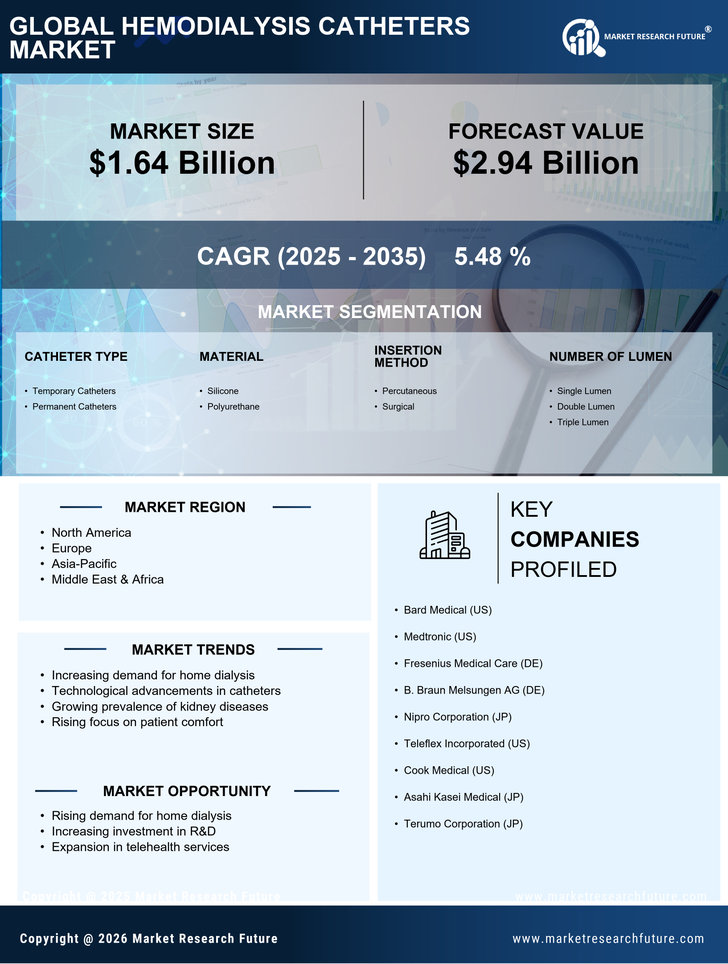
Healthcare Market Segmentation
Healthcare By Catheter Type (USD Billion, 2025-2035)
- Temporary Catheters
- Permanent Catheters
Healthcare By Material (USD Billion, 2025-2035)
- Silicone
- Polyurethane
Healthcare By Insertion Method (USD Billion, 2025-2035)
- Percutaneous
- Surgical
Healthcare By Number of Lumen (USD Billion, 2025-2035)
- Single Lumen
- Double Lumen
- Triple Lumen
Healthcare By End User (USD Billion, 2025-2035)
- Hospitals
- Home Care Settings
- Dialysis Centers