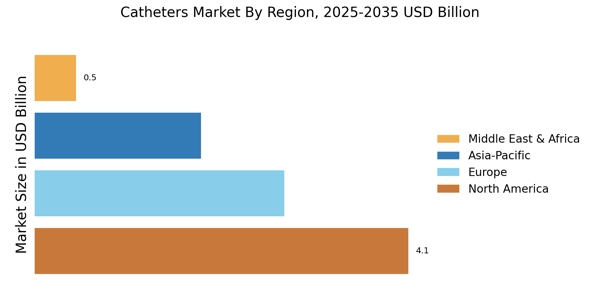
Focus on Patient Safety in Catheters Market
Patient safety remains a paramount concern within the Catheters Market, influencing product development and regulatory standards. Manufacturers are increasingly prioritizing the design of catheters that minimize the risk of infections and other complications. The implementation of safety features, such as antimicrobial coatings and advanced sealing mechanisms, is becoming commonplace.
Regulatory bodies are also tightening guidelines to ensure that catheters meet stringent safety criteria. This focus on patient safety is likely to drive innovation and investment in the market, as healthcare providers seek to adopt products that enhance patient care. The emphasis on safety is expected to contribute to a steady growth trajectory for the Catheters Market.
Technological Advancements in Catheters Market
The Catheters Market is experiencing a notable transformation due to rapid technological advancements. Innovations such as smart catheters equipped with sensors and real-time monitoring capabilities are enhancing patient outcomes. These advancements not only improve the accuracy of catheter placements but also reduce the risk of complications.
The integration of materials science has led to the development of catheters that are more biocompatible and durable, which is crucial for long-term use. Furthermore, the introduction of 3D printing technology allows for the customization of catheters, catering to specific patient needs. As a result, the market is projected to grow significantly, with estimates suggesting a compound annual growth rate of over 8% in the coming years.
Aging Population and Chronic Diseases Impacting Catheters Market
The Catheters Market is significantly influenced by the aging population and the rising prevalence of chronic diseases. As the global demographic shifts towards an older population, the demand for medical devices, including catheters, is expected to increase. Older adults are more susceptible to conditions such as cardiovascular diseases, diabetes, and urinary incontinence, necessitating catheter use for management and treatment.
Market analysis suggests that the segment catering to chronic disease management will account for a substantial share of the overall catheter market. This demographic trend is likely to drive sustained growth in the Catheters Market, as healthcare systems adapt to meet the needs of an aging population.
Rising Demand for Minimally Invasive Procedures in Catheters Market
The Catheters Market is witnessing a surge in demand for minimally invasive procedures, driven by the increasing preference for less traumatic surgical options. Patients and healthcare providers alike are gravitating towards techniques that promise quicker recovery times and reduced hospital stays.
This trend is particularly evident in fields such as cardiology and urology, where catheter-based interventions are becoming the standard. The market data indicates that the revenue generated from minimally invasive catheter procedures is expected to reach several billion dollars by 2026. This shift not only enhances patient satisfaction but also reduces healthcare costs, thereby propelling the growth of the Catheters Market.
Increasing Healthcare Expenditure and Investment in Catheters Market
The Catheters Market is benefiting from increasing healthcare expenditure and investment in medical technologies. Governments and private sectors are allocating more resources towards healthcare infrastructure, which includes the procurement of advanced medical devices.
This trend is particularly evident in emerging economies, where healthcare systems are rapidly evolving. The investment in healthcare is expected to enhance access to catheter-based treatments, thereby expanding the market. Furthermore, as healthcare providers seek to improve patient outcomes, the demand for innovative catheter solutions is likely to rise. Market forecasts indicate that this increase in healthcare spending will play a crucial role in propelling the growth of the Catheters Market.