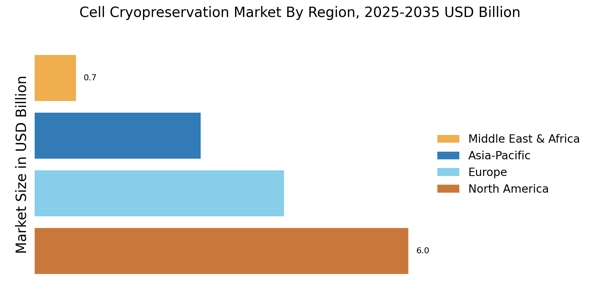
The Cell Cryopreservation Market is currently experiencing a notable evolution, driven by advancements in biobanking and regenerative medicine. As the demand for preserving cellular materials for research and therapeutic applications continues to rise, various stakeholders are increasingly investing in innovative technologies.3
This trend appears to be fueled by the growing recognition of the importance of cellular preservation in enhancing the efficacy of treatments and ensuring the viability of biological samples over extended periods. Furthermore, the integration of automation and artificial intelligence in cryopreservation processes is likely to streamline operations, reduce human error, and improve overall efficiency.
In addition, the Cell Cryopreservation Market is witnessing a shift towards personalized medicine, which emphasizes tailored treatment approaches based on individual patient profiles. This paradigm shift may lead to an increased focus on the preservation of specific cell types, such as stem cells, which hold immense potential for regenerative therapies.
Moreover, regulatory frameworks are evolving to support the safe and effective use of cryopreserved materials, thereby fostering a conducive environment for market growth. As the landscape continues to change, stakeholders must remain vigilant and adaptable to emerging trends and technologies that could shape the future of cellular preservation.
Technological Advancements
The Cell Cryopreservation Market is increasingly influenced by technological innovations that enhance preservation techniques. Developments in cryoprotectants and freezing protocols are likely to improve cell viability and functionality post-thawing. Furthermore, automation in cryopreservation processes may lead to greater consistency and efficiency, reducing the risk of human error.
Advancements in cryopreservation technologies are strongly supported by increasing global healthcare innovation and disease monitoring needs. The European Centre for Disease Prevention and Control highlights over 4 million infectious disease cases monitored annually in Europe, driving demand for high-quality biological sample storage. Additionally, the Pan American Health Organization emphasizes strengthening laboratory infrastructure across Latin America, boosting adoption of automated and advanced cryopreservation systems for improved sample integrity and research accuracy.
Personalized Medicine
A growing emphasis on personalized medicine is shaping the Cell Cryopreservation Market. This trend suggests that there will be a heightened focus on preserving specific cell types, such as stem cells, to cater to individualized treatment plans. As healthcare moves towards more tailored approaches, the demand for specialized specialized stem cell freezing media, cryopreservation solutions and the consistent purchase DMSO may increase.
The shift toward personalized medicine is reinforced by increasing investments in targeted therapies and reproductive health. The United Nations Population Fund reports that over 121 million unintended pregnancies occur annually, increasing demand for fertility preservation such as oocyte cryopreservation. Furthermore, UNICEF highlights expanding newborn screening programs globally, supporting early-stage biological sample storage and individualized treatment planning, thereby accelerating demand for specialized cryopreservation media and stem cell preservation solutions.
Regulatory Developments
The evolving regulatory landscape surrounding the Cell Cryopreservation Market indicates a potential for increased oversight and standardization. As regulations adapt to the advancements in cryopreservation technologies, stakeholders may find opportunities for growth while ensuring compliance with safety and efficacy standards.
Evolving regulatory frameworks are enhancing safety and standardization in cryopreservation practices globally. The Global Fund to Fight AIDS, Tuberculosis and Malaria has invested over USD 60 billion in health programs, strengthening laboratory systems and compliance standards. Meanwhile, Médecins Sans Frontières supports advanced cold-chain and storage protocols in over 70 countries, reinforcing the need for regulated, high-quality cryopreservation infrastructure aligned with global healthcare standards.