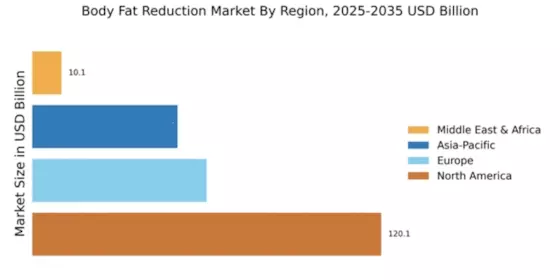
North America: Expanding body fat reduction
The regulatory framework for the body fat reduction market in North America is shaped by both national health authorities and industry standards, with distinct approaches in the United States and Canada. In the U.S., surgical procedures like liposuction are regulated under strict guidelines, particularly when performed under anything beyond local anesthesia. These procedures must be carried out in accredited facilities, recognized by organizations such as the AAAASF, AAAHC, AOS, or JCAHO, ensuring patient safety and procedural quality. Non-invasive body sculpting treatments, however, are not directly regulated by specific federal laws, though the devices used may fall under FDA oversight depending on their intended use and medical claims. In contrast, Canada adopts a more nuanced regulatory framework through Health Canada, focusing on how a device or product functions and the claims associated with it, regardless of whether the treatment is surgical or non-surgical. This leads to a sophisticated classification system that carefully evaluates devices and products that fall between cosmetic and medical use. For pharmaceutical approaches to fat reduction and obesity, both countries require comprehensive clinical trial data to prove safety and efficacy.
Europe: Strong Production body fat products
In Europe, the regulatory framework for the body fat reduction market has become significantly more stringent under the Medical Device Regulation (MDR) (EU) 2017/745, which replaced the older Medical Devices Directive (MDD). Unlike the MDD, the MDR explicitly includes certain aesthetic and cosmetic devices, even those without a medical purpose, under its scope, as outlined in Annex XVI. This includes body contouring and laser fat reduction devices, which must now comply with rigorous safety, clinical evidence, and performance requirements to obtain CE marking for market access. This shift reflects a growing recognition of the potential health risks associated with aesthetic procedures, even when they are not intended to treat medical conditions. On the pharmaceutical side, the European Medicines Agency (EMA) oversees the regulation of anti-obesity drugs across EU member states. Approval requires extensive clinical trial data to demonstrate safety and efficacy. Approved drugs include liraglutide and naltrexone/bupropion, though access may vary due to differing reimbursement policies across the region. Post-Brexit, the UK’s Medicines and Healthcare products Regulatory Agency (MHRA) functions independently but continues to maintain standards closely aligned with the EMA.
Asia-Pacific : Rapidly Growing Market
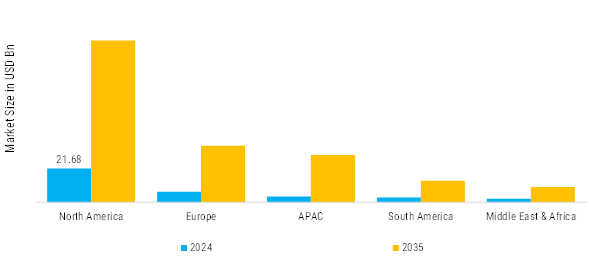
The Asia-Pacific region is emerging as a significant player in the body fat reduction market, with a projected size of 40.0 million by 2025. Countries like China, Japan, and India are key contributors, while the South Korea body fat reduction market is also gaining attention due to the country’s strong cosmetic surgery and aesthetic treatment industry. Governments are implementing policies to combat obesity, which is further driving demand for body fat reduction solutions. The region's diverse population presents unique challenges and opportunities for tailored health solutions, making it a dynamic market. The competitive landscape is characterized by both local and international players, including companies like Eli Lilly and Company and Amgen. As the market matures, there is a noticeable shift towards innovative and personalized solutions, catering to the specific needs of consumers in this diverse region.
South America: Growing clinical quality
The regulatory framework for the body fat reduction market in South America is anchored in a growing emphasis on patient safety, clinical quality, and professional accountability, though the level of oversight varies significantly from country to country. Accreditation serves as a baseline for safety and quality, setting minimum standards that clinics and hospitals must meet to operate legally, particularly in procedures like liposuction and other fat reduction treatments. Colombia leads the region in regulatory rigor, with its Ministry of Health and the Colombian Society of Plastic Surgery (SCCP) working in tandem to enforce strict rules around surgeon qualifications, hygiene protocols, and patient care standards. Clinics in Colombia must demonstrate ongoing compliance, including continuous professional development and operational safety, or risk closure. The SCCP plays a critical role by ensuring that only board-certified plastic surgeons perform invasive procedures such as liposuction.
Middle East & Africa: Emerging stainless essentials
In Middle East & Africa, the landscape is less uniform. Most countries still lack clear or enforceable regulations for cosmetic procedures like liposuction, creating significant safety concerns. South Africa is an exception, with a relatively advanced framework. Bodies like APRASSA set eligibility guidelines for patients undergoing fat reduction procedures, and institutions such as Mediclinic work with certified surgeons and offer structured treatment plans. However, in many other African nations, poor enforcement and regulatory gaps allow unlicensed practitioners and unregulated clinics to operate freely. While the Middle East is moving toward harmonized, EU-aligned safety standards, Africa’s fat reduction market still requires stronger institutional oversight to ensure patient safety and procedural quality. Additionally, The South African Health Products Regulatory Authority (SAHPRA) regulates anti-obesity drugs, requiring clinical trial data. The market is small but growing, driven by increasing obesity rates and awareness. Access to newer therapies is limited due to cost and regulatory hurdles.