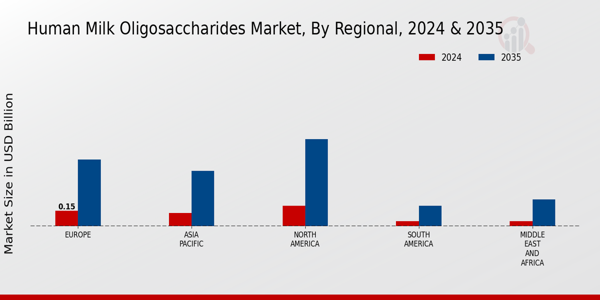
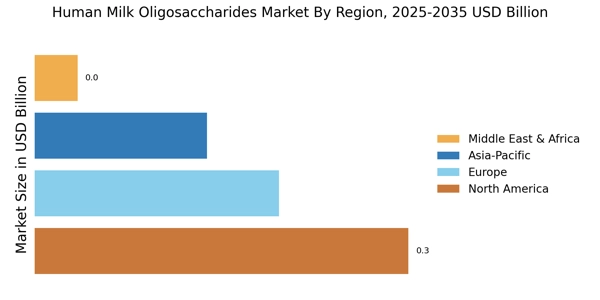Global market valuation was derived through revenue mapping and volume consumption analysis. The methodology included:
Identification of 35+ key manufacturers and biotechnology firms across North America, Europe, Asia-Pacific, and Latin America specializing in 2'-FL, 3'-FL, LNT, and complex HMO production
Product mapping across infant formula, functional foods, dietary supplements, and clinical nutrition categories
Technology assessment covering microbial fermentation (bacterial/yeast), enzymatic synthesis, and human milk-derived sources
Analysis of reported and modeled annual revenues specific to HMO portfolios and ingredient sourcing
Coverage of manufacturers representing 75-80% of global market share in 2024
Extrapolation using bottom-up (volume consumption × ASP by country/region) and top-down (manufacturer revenue validation) approaches to derive segment-specific valuations for powder, liquid, and capsule formulations across healthcare, F&B, and nutraceutical end-use sectors
Key Differences from Your Template:
Regulatory sources shifted from aesthetic medical devices to food/infant nutrition authorities (EFSA, Codex Alimentarius, FSANZ)
Scientific sources focused on pediatric nutrition, microbiome research, and lactation science rather than surgical societies
Primary research adjusted percentages to reflect the HMO industry's structure (more balanced Tier 1/Tier 2, stronger European regulatory focus, emphasis on Director-level technical contacts)
Market estimation adjusted for biotechnology/specialty ingredient company landscape vs. medical device manufacturers