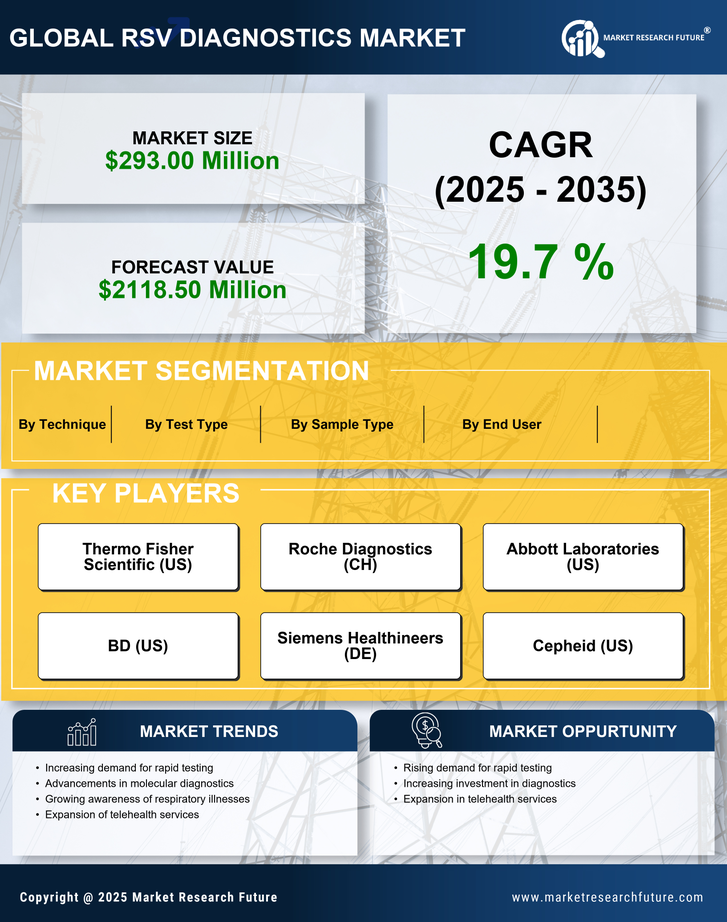
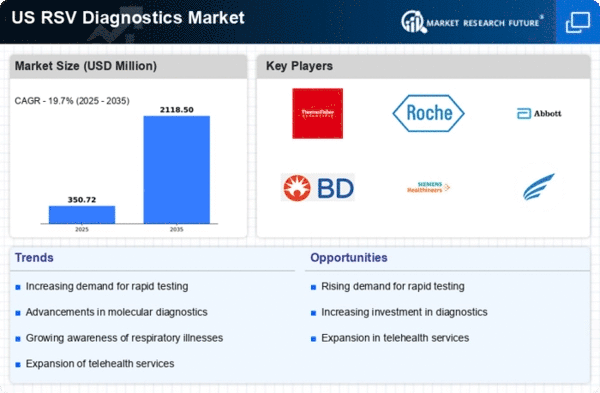
US Pharmaceutical Market Segmentation
Pharmaceutical By Test Type (USD Million, 2025-2035)
- Molecular Tests
- Antigen Tests
- Serological Tests
- Virus Culture
Pharmaceutical By End User (USD Million, 2025-2035)
- Hospitals
- Diagnostic Laboratories
- Research Institutions
Pharmaceutical By Sample Type (USD Million, 2025-2035)
- Nasal Swabs
- Throat Swabs
- Bronchoalveolar Lavage
Pharmaceutical By Technique (USD Million, 2025-2035)
- Polymerase Chain Reaction
- Enzyme-Linked Immunosorbent Assay
- Immunofluorescence Assay