North America : Market Leader in Diagnostics
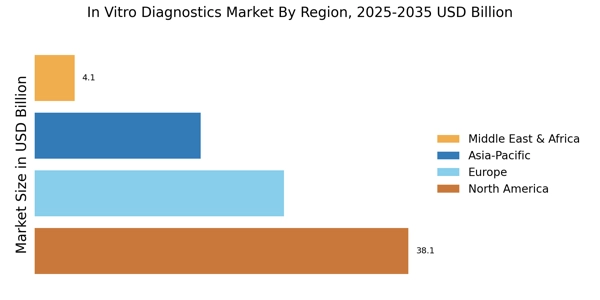
North America leads in the In Vitro Diagnostics Market Size, accounting for over 38.1% of the global revenue in 2024. Key growth drivers include advanced healthcare infrastructure, increasing prevalence of chronic diseases, and a strong focus on personalized medicine. Regulatory support from agencies like the FDA further catalyzes innovation and market expansion.
- CDC reports over 1 billion laboratory tests conducted annually in the U.S., while NIH funding exceeds USD 45 billion per year, and WHO highlights widespread chronic disease prevalence, supporting North America’s leadership with over 38.1% market share.
The US market is the primary contributor, followed by Canada, which is experiencing significant growth due to rising healthcare investments. The competitive landscape is characterized by the presence of major players such as Abbott Laboratories, Thermo Fisher Scientific, and Roche Diagnostics. These companies are investing heavily in R&D to develop innovative diagnostic solutions. The market is also witnessing a trend towards point-of-care testing, driven by the need for rapid and accurate results. This shift is expected to enhance patient outcomes and streamline healthcare delivery.
Europe : Regulatory Framework and Innovation
Europe In Vitro Diagnostics Market size was valued at USD 25.38 billion in 2024, making it the second-largest regional market with 30% share. The region's growth is driven by an aging population, increasing healthcare expenditure, and a robust regulatory framework that encourages innovation.
- WHO Europe reports over 25% population aged 60+, while ECDC highlights increasing diagnostic testing across EU nations. With 30% market share and USD 25.38 billion valuation, Europe shows strong demand for advanced in vitro diagnostic technologies.
The European Union's In Vitro Diagnostic Regulation (IVDR) aims to enhance patient safety and ensure high-quality diagnostic products, further stimulating market growth. Leading countries in this region include Germany, France, and the UK, which are home to several key players like Siemens Healthineers and bioMérieux.
The competitive landscape is marked by a strong emphasis on technological advancements and partnerships among companies to enhance product offerings. The presence of well-established healthcare systems facilitates the adoption of innovative diagnostic solutions, making Europe a vital hub for market expansion.
Asia-Pacific : Emerging Market Potential
Asia-Pacific is rapidly emerging as a significant player in the In Vitro Diagnostics Market, holding approximately 20% of the global share. The region's growth is fueled by increasing healthcare investments, rising awareness of preventive healthcare, and a growing population with chronic diseases. Countries like China and India are leading this growth, supported by government initiatives aimed at improving healthcare access and quality. The competitive landscape in Asia-Pacific is evolving, with both multinational corporations and local players vying for market share.
Key players such as Danaher Corporation and Becton Dickinson are expanding their presence through strategic partnerships and acquisitions. The region is also witnessing a surge in demand for point-of-care testing, driven by the need for rapid diagnostics in remote areas, further enhancing market dynamics.
Middle East and Africa : Untapped Market Opportunities
The Middle East and Africa represent an emerging market, holding approximately 5% of the global share. The growth in this region is driven by increasing healthcare investments, rising prevalence of infectious diseases, and a growing focus on improving healthcare infrastructure. Ecosystems like South Africa and the UAE are leading the market, supported by government initiatives aimed at enhancing healthcare access and quality.
The competitive landscape is characterized by a mix of local and international players, with in vitro diagnostics companies like Ortho Clinical Diagnostics and Hologic, Inc. making significant inroads. The region is witnessing a growing demand for innovative diagnostic solutions, particularly in point-of-care testing, which is crucial for addressing healthcare challenges in remote and underserved areas. This presents significant opportunities for IVD industry expansion and investment.