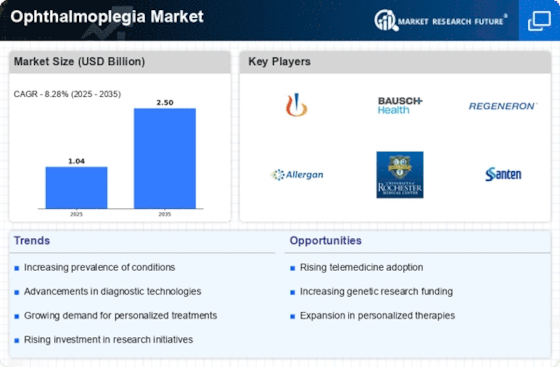
Healthcare Market Segmentation
Healthcare By Type of Ophthalmoplegia (USD Billion, 2025-2035)
- Congenital Ophthalmoplegia
- Acquired Ophthalmoplegia
- Botulism Ophthalmoplegia
- Thyroid Eye Disease
Healthcare By Symptoms (USD Billion, 2025-2035)
- Diplopia
- Ptosis
- Strabismus
- Limited Eye Movement
Healthcare By Treatment Type (USD Billion, 2025-2035)
- Surgical Intervention
- Medication
- Physical Therapy
Healthcare By End User (USD Billion, 2025-2035)
- Hospitals
- Ophthalmology Clinics
- Home Care Settings