North America : Leading Innovation and Standards
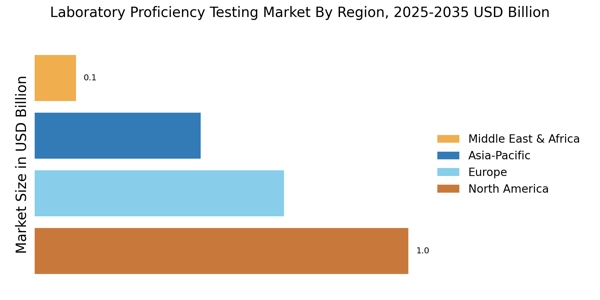
North America held the largest Laboratory Proficiency Testing Market Share, accounting for over 45.33% of the global market in 2024. The region's growth is driven by stringent regulatory requirements, increasing laboratory accreditation, and a rising focus on quality assurance in healthcare. The demand for proficiency testing is further fueled by advancements in technology and the growing number of laboratories seeking to enhance their operational standards.
- The Centers for Disease Control and Prevention (CDC) reports billions of laboratory tests are performed annually in the U.S., while the Pan American Health Organization (PAHO) highlights that over 90% of healthcare facilities in the Americas utilize standardized diagnostic systems, reinforcing strong demand for proficiency testing and quality assurance programs.
The United States and Canada are the leading countries in this market, with the U.S. accounting for the majority of the market share. Key players such as Bio-Rad Laboratories, Inc. and LGC Limited are prominent in this region, providing a wide range of proficiency testing solutions. The competitive landscape is characterized by continuous innovation and partnerships among leading organizations to enhance service offerings.
Europe : Regulatory Framework and Growth
Europe Laboratory Proficiency Testing Market size was valued at USD 0.66 billion in 2024, making it the second-largest regional market with a 30% share. The region benefits from robust regulatory frameworks, such as the In Vitro Diagnostic Regulation (IVDR), which mandates proficiency testing for laboratories. This regulatory environment, combined with an increasing emphasis on quality control in healthcare, drives demand for proficiency testing services across various sectors.
- The European Centre for Disease Prevention and Control (ECDC) coordinates surveillance systems across 30 EU/EEA countries, while the World Health Organization (WHO) reports that Europe conducts millions of diagnostic tests annually, supporting strong regulatory compliance and continuous demand for laboratory proficiency testing services.
Germany, the United Kingdom, and France are the leading countries in this market, with Germany being the largest contributor. The competitive landscape features key players like Merck KGaA and Randox Laboratories Ltd., who are actively involved in enhancing proficiency testing standards. The presence of numerous accredited laboratories further supports market growth, fostering a culture of quality assurance and compliance.
Asia-Pacific : Emerging Markets and Opportunities
Asia-Pacific is witnessing significant growth in the laboratory proficiency testing market, holding approximately 20% of the global share. The region's expansion is driven by increasing healthcare investments, a growing number of diagnostic laboratories, and rising awareness of quality standards. Countries like China and India are leading this growth, supported by government initiatives aimed at improving healthcare infrastructure and laboratory capabilities.
China is the largest market in the region, followed by India and Japan. The competitive landscape is evolving, with both local and international players, such as Eurofins Scientific and Hach Company, expanding their presence. The demand for proficiency testing is expected to rise as laboratories seek to comply with international standards and enhance their service quality, creating ample opportunities for growth in this sector.
Middle East and Africa : Growing Focus on Quality Standards
The Middle East and Africa region is gradually emerging in the laboratory proficiency testing market, accounting for approximately 5% of the global share. The growth is primarily driven by increasing healthcare investments, rising laboratory establishments, and a growing emphasis on quality assurance in diagnostics. Countries like South Africa and the UAE are at the forefront, with initiatives aimed at enhancing laboratory standards and accreditation processes.
South Africa is the leading market in this region, with a growing number of accredited laboratories seeking proficiency testing services. The competitive landscape is characterized by the presence of both local and international players, including Silliker, Inc. and Intertek Group plc. As the region continues to develop its healthcare infrastructure, the demand for proficiency testing is expected to rise, fostering a culture of quality and compliance in laboratory practices.