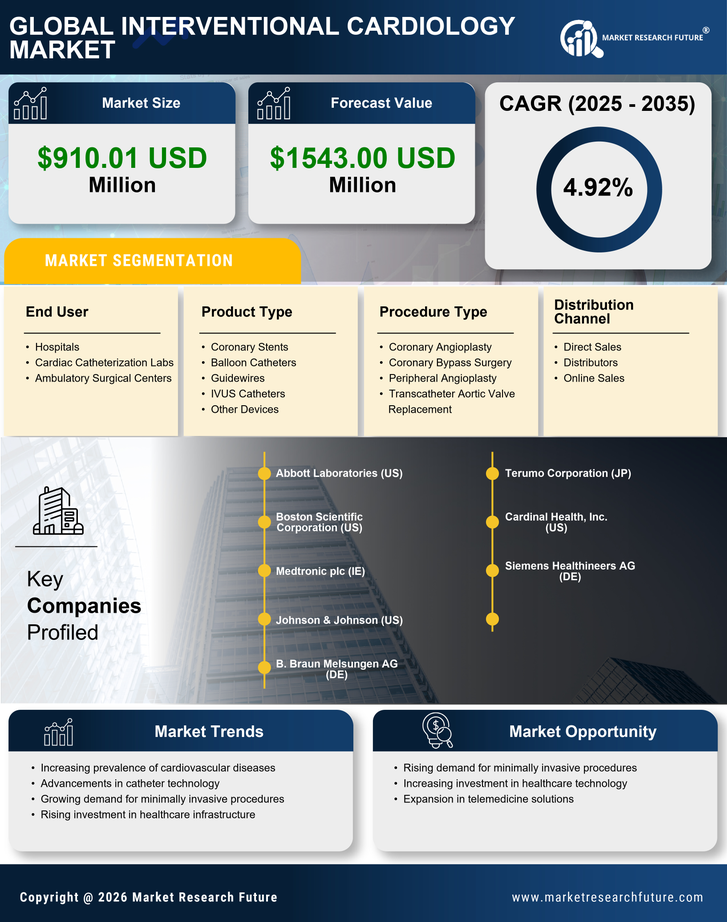
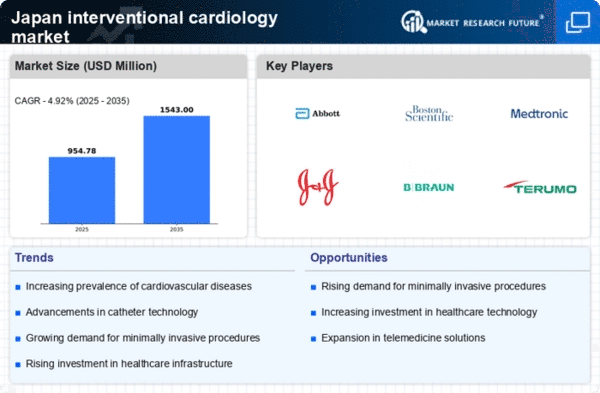
Japan Security, Access Control and Robotics Market Segmentation
Security, Access Control and Robotics By Product Type (USD Million, 2025-2035)
- Coronary Stents
- Balloon Catheters
- Guidewires
- IVUS Catheters
- Other Devices
Security, Access Control and Robotics By Procedure Type (USD Million, 2025-2035)
- Coronary Angioplasty
- Coronary Bypass Surgery
- Peripheral Angioplasty
- Transcatheter Aortic Valve Replacement
Security, Access Control and Robotics By End User (USD Million, 2025-2035)
- Hospitals
- Cardiac Catheterization Labs
- Ambulatory Surgical Centers
Security, Access Control and Robotics By Distribution Channel (USD Million, 2025-2035)
- Direct Sales
- Distributors
- Online Sales