
Influenza Diagnostics Market
Influenza Diagnostics Market Summary
As per MRFR analysis, the Influenza Diagnostics Market Size was estimated at 9.26 USD Billion in 2024. The Influenza Diagnostics industry is projected to grow from 10.14 USD Billion in 2025 to 22.43 USD Billion by 2035, exhibiting a compound annual growth rate (CAGR) of 8.33% during the forecast period 2025 - 2035.
Key Market Trends & Highlights
The Influenza Diagnostics Market is poised for substantial growth driven by technological advancements and increasing demand for rapid testing solutions.
- Technological advancements in diagnostics are reshaping the landscape of influenza testing, particularly in North America.
- The demand for rapid diagnostic tests remains robust, making it the largest segment in the market.
- Preventive healthcare is gaining traction, with a notable emphasis on early detection and management of influenza infections in the Asia-Pacific region.
- Key market drivers include rising incidence of influenza infections and government initiatives aimed at enhancing diagnostic capabilities.
- Rapid Influenza Diagnostic Tests (RIDTs) currently hold a significant market share due to their quick turnaround time, ease of use, and suitability for point-of-care settings.
Market Size & Forecast
| 2024 Market Size | 9.26 (USD Billion) |
| 2035 Market Size | 22.43 (USD Billion) |
| CAGR (2025 - 2035) | 8.33% |
Major Players
Roche Diagnostics(CH), Abbott Laboratories (US), Thermo Fisher Scientific (US), BD (Becton, Dickinson and Company) (US), Siemens Healthineers (DE), Cepheid (US), Hologic (US), Quidel Corporation (US)
Influenza Diagnostics Market Trends
The Influenza Diagnostics Market is currently experiencing a dynamic evolution, driven by advancements in technology and an increasing emphasis on rapid and accurate diagnostic methods within the influenza diagnostic market. The growing prevalence of influenza outbreaks globally has heightened the demand for effective diagnostic solutions. As healthcare providers seek to enhance patient outcomes, the market is witnessing a shift towards innovative testing methodologies, including molecular diagnostics and point-of-care testing in the influenza diagnostic tests market. These developments not only facilitate timely diagnosis but also contribute to better management of influenza cases, thereby reducing the burden on healthcare systems.
Moreover, the rising awareness regarding the importance of early detection and treatment of influenza is propelling influenza diagnostics market growth. Stakeholders are increasingly investing in research and development to create more efficient diagnostic tools. This trend is further supported by collaborations between public health organizations and private entities, aimed at improving surveillance and response strategies. As the Influenza Diagnostics Market continues to expand, it appears poised to play a crucial role in global health initiatives, particularly in enhancing preparedness for seasonal epidemics and potential pandemics, as highlighted in various influenza diagnostics market analysis studies.
Technological Advancements in Diagnostics
The Influenza Diagnostics Market is witnessing a surge in technological innovations, particularly in molecular diagnostics. These advancements enable faster and more accurate detection of influenza viruses, which is essential for effective patient management and strengthening the influenza diagnostics market landscape. The integration of artificial intelligence and machine learning into diagnostic processes is also emerging, potentially enhancing the precision of test results.
Shift Towards Point-of-Care Testing
There is a notable trend towards point-of-care testing within the Influenza Diagnostics Market. This approach allows for immediate results at the site of care, reducing the time between testing and treatment initiation. Such convenience is likely to improve patient outcomes and streamline healthcare delivery, particularly in remote or underserved areas, supporting the broader influenza diagnostic tests market expansion.
Increased Focus on Preventive Healthcare
The growing emphasis on preventive healthcare is influencing the Influenza Diagnostics Market. Stakeholders are prioritizing early detection and vaccination strategies to mitigate the impact of influenza outbreaks. This trend suggests a shift in healthcare paradigms, where proactive measures are increasingly recognized as vital components of public health.
Influenza Diagnostics Market Drivers
Market Growth Projections
The Global Influenza Diagnostics Market Industry is poised for substantial growth, with projections indicating a market size of 7.22 USD Billion in 2024 and an anticipated increase to 11.9 USD Billion by 2035. This growth trajectory reflects a compound annual growth rate of 4.69% from 2025 to 2035. Such figures suggest a robust demand for influenza diagnostic solutions, driven by factors such as rising incidences of influenza, technological advancements, and increased healthcare access. The market's expansion is indicative of the critical role that effective diagnostics play in managing influenza outbreaks and protecting public health.
Rising Incidence of Influenza
The Global Influenza Diagnostics Market Industry is experiencing growth due to the increasing incidence of influenza infections worldwide. Seasonal outbreaks and pandemics contribute to a heightened demand for diagnostic tools. For instance, the World Health Organization indicates that influenza affects millions of people annually, leading to significant morbidity and mortality. This rising incidence necessitates accurate and timely diagnostics, thereby driving the market. The market is projected to reach 7.22 USD Billion in 2024, reflecting the urgent need for effective influenza diagnostics to manage public health challenges.
Government Initiatives and Funding
Government initiatives aimed at improving public health infrastructure significantly influence the Global Influenza Diagnostics Market Industry. Increased funding for influenza surveillance and research programs enhances the development and distribution of diagnostic tools. For instance, various health departments worldwide allocate resources to combat influenza through vaccination campaigns and diagnostic improvements. These initiatives not only bolster the market but also ensure that healthcare systems are better equipped to handle influenza outbreaks. The anticipated compound annual growth rate of 4.69% from 2025 to 2035 underscores the positive impact of such government efforts on market growth.
Growing Awareness of Influenza Prevention
The Global Influenza Diagnostics Market Industry benefits from the growing awareness surrounding influenza prevention and control. Public health campaigns emphasize the importance of early diagnosis and treatment, leading to increased demand for diagnostic tests. Educational initiatives by health organizations aim to inform the public about the symptoms and risks associated with influenza, thereby promoting proactive healthcare behaviors. This heightened awareness contributes to a more informed population that seeks timely diagnostic solutions, further propelling market growth. As awareness continues to rise, the market is expected to expand in response to the demand for effective diagnostic tools.
Technological Advancements in Diagnostics
Technological innovations play a pivotal role in the Global Influenza Diagnostics Market Industry. The development of rapid diagnostic tests, including molecular assays and point-of-care testing, enhances the speed and accuracy of influenza detection. These advancements facilitate timely treatment and containment of outbreaks. For example, the introduction of multiplex PCR assays allows for the simultaneous detection of multiple respiratory pathogens, improving diagnostic efficiency. As technology continues to evolve, the market is likely to expand, with projections indicating a growth to 11.9 USD Billion by 2035, driven by the demand for advanced diagnostic solutions.
Emerging Markets and Increased Healthcare Access
Emerging markets are becoming increasingly significant in the Global Influenza Diagnostics Market Industry. As healthcare access improves in developing regions, the demand for influenza diagnostics rises correspondingly. Countries in Asia and Africa are witnessing investments in healthcare infrastructure, leading to enhanced diagnostic capabilities. This trend is crucial as it allows for better disease management and surveillance, ultimately reducing the burden of influenza. The growth potential in these markets is substantial, with the overall market expected to grow steadily, driven by the increasing accessibility of diagnostic tools and services.
Market Segment Insights
By Application: Diagnostic Testing (Largest) vs. Surveillance (Fastest-Growing)
The Influenza Diagnostics Market showcases a diverse array of application segments, with Diagnostic Testing leading in market share. This segment dominates due to its critical role in identifying and diagnosing influenza infections effectively. Surveillance is an emerging component, gaining traction as health authorities emphasize tracking influenza trends to manage outbreaks efficiently. The focus on public health and real-time data collection is propelling surveillance figures upward, making it a key player in the diagnostics landscape. Growth trends in the application segment are primarily driven by heightened awareness of influenza's impact on public health and advancements in diagnostic technologies. The shift towards rapid and accurate testing methods is fostering innovations in diagnostic testing, while the need for robust surveillance systems is fueled by global health challenges. Continuous investments in Research and Development are also enhancing the efficiency of diagnostics, ensuring that both existing and emerging segments thrive in the overall market.
Diagnostic Testing (Dominant) vs. Surveillance (Emerging)
The Diagnostic Testing segment in the Influenza Diagnostics Market stands as the dominant force, characterized by a wide array of testing methodologies and technologies that facilitate accurate and timely diagnosis of influenza. Its significance is underscored by the increasing prevalence of flu viruses and the need for reliable testing solutions. Conversely, the Surveillance segment is emerging as a vital player, with a growing emphasis on real-time monitoring of influenza trends to effectively guide public health responses. Both segments are essential, with Diagnostic Testing highlighting established methodologies while Surveillance capitalizes on innovative data collection practices to enhance epidemic preparedness and response.
By Test Type: Molecular Tests (Largest) vs. Antigen Tests (Fastest-Growing)
In the Influenza Diagnostics Market, molecular tests have secured the largest market share, driven by their high accuracy and ability to detect the virus at an early stage. Antigen tests, while typically associated with lower sensitivity, have gained traction rapidly due to their quick turnaround times and ease of use, particularly in point-of-care settings. Other segments such as serological tests, rapid diagnostic tests, and culture tests comprise smaller portions of the market but serve crucial roles in specific applications like epidemiological studies and confirming diagnoses. As the market evolves, antigen tests are experiencing the fastest growth, largely influenced by increasing demand for rapid testing solutions, especially in pandemic scenarios. Advances in technology are making these tests more reliable and easier to administer. Meanwhile, molecular tests continue to benefit from ongoing innovation, solidifying their role as the gold standard for influenza diagnosis. Both test categories are expected to witness shifts in market dynamics, influenced by regulatory changes and healthcare policies that favor faster and more efficient diagnostics.
Molecular Tests (Dominant) vs. Rapid Diagnostic Tests (Emerging)
Molecular tests are characterized by their high sensitivity and specificity, making them the preferred choice for clinical laboratories in diagnosing influenza. This dominant position is supported by technological advancements that continuously enhance their accuracy and usability. In contrast, rapid diagnostic tests represent an emerging segment that caters to the growing need for immediate results in various healthcare environments, including emergency and primary care settings. Although these tests may not reach the same accuracy levels as molecular tests, their convenience and speed facilitate timely clinical decisions. The balance between these two test types reflects a broader trend towards immediate patient care while still affirming the importance of reliable diagnostic methodologies in managing influenza outbreaks.
By End User: Hospitals (Largest) vs. Diagnostic Laboratories (Fastest-Growing)
In the Influenza Diagnostics Market, hospitals capture the most substantial share among the end users, due to their extensive services and access to advanced diagnostic technologies. Diagnostic laboratories follow closely, benefiting from the rising demand for specialized testing services, which enables them to enhance their market presence. Research institutions and home care settings are also crucial players but currently hold smaller portions of the market, reflecting a more niche approach to influenza diagnosis.
Hospitals (Dominant) vs. Research Institutions (Emerging)
Hospitals serve as the dominant end user in the influenza diagnostics market, equipped with sophisticated technology and a consistent flow of patients requiring accurate diagnosis and treatment. Their established infrastructure allows for rapid testing and results, thereby supporting effective patient management. On the other hand, research institutions are emerging players, focusing on the development of novel diagnostic methods and greater understanding of influenza viruses. Although their market share is smaller, they play a pivotal role in innovating testing solutions, helping to drive advancements in the field that may transform future clinical practices.
Regional Insights
North America : Market Leader in Diagnostics
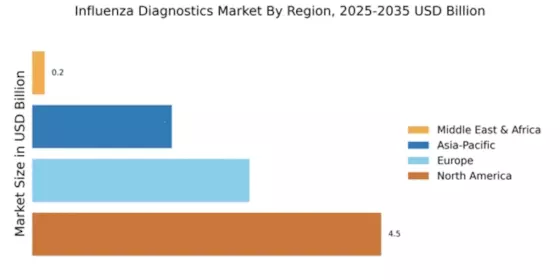
North America is poised to maintain its leadership in the Influenza Diagnostics Market, holding a significant market share of 4.5 in 2024. The region's growth is driven by increasing healthcare expenditure, advanced diagnostic technologies, and a robust regulatory framework that encourages innovation. The rising incidence of influenza and the demand for rapid diagnostic tests further fuel market expansion. Regulatory bodies are actively promoting the adoption of new diagnostic methods, enhancing the overall market landscape. The competitive landscape in North America is characterized by the presence of major players such as Roche Diagnostics, Abbott Laboratories, and Thermo Fisher Scientific. These companies are investing heavily in research and development to introduce innovative diagnostic solutions. The U.S. remains the largest market, supported by a well-established healthcare infrastructure and a high prevalence of respiratory diseases. The focus on improving patient outcomes and reducing healthcare costs is driving the demand for efficient diagnostic tools.
Europe : Emerging Market Dynamics
Europe is witnessing a growing demand for influenza diagnostics, with a market size of 2.8 in 2024. The region's growth is propelled by increasing awareness of influenza prevention and the implementation of stringent health regulations. The European Union's commitment to enhancing public health through improved diagnostic capabilities is a key driver. Additionally, the rise in seasonal influenza outbreaks has led to a surge in testing, further boosting market growth. Leading countries in Europe include Germany, France, and the UK, where major players like Siemens Healthineers and Hologic are actively expanding their market presence. The competitive landscape is marked by collaborations and partnerships aimed at developing advanced diagnostic solutions. Regulatory bodies are emphasizing the importance of timely and accurate diagnostics, as highlighted by the European Centre for Disease Prevention and Control, which states that "effective influenza surveillance is crucial for public health management."
Asia-Pacific : Rapidly Growing Market
The Asia-Pacific region is emerging as a significant player in the Influenza Diagnostics Market, with a market size of 1.8 in 2024. The growth is driven by increasing healthcare investments, rising awareness of infectious diseases, and the need for rapid diagnostic solutions. Governments are implementing policies to enhance healthcare infrastructure, which is expected to further stimulate market growth. The region's diverse population and varying healthcare needs present unique opportunities for diagnostic innovations. Countries like China, India, and Japan are leading the market, with a growing number of local and international players entering the space. Companies such as BD and Quidel Corporation are expanding their operations in the region. The competitive landscape is evolving, with a focus on affordability and accessibility of diagnostic tests. However, challenges such as regulatory hurdles and varying healthcare standards remain prevalent, impacting market dynamics.
Middle East and Africa : Emerging Healthcare Landscape
The Middle East and Africa (MEA) region is gradually developing its influenza diagnostics market, currently valued at 0.16 in 2024. The growth is primarily driven by increasing healthcare investments and the rising burden of infectious diseases. Governments are focusing on improving healthcare infrastructure and access to diagnostic services, which is essential for managing influenza outbreaks. The region's diverse healthcare challenges necessitate tailored diagnostic solutions to meet local needs. Countries such as South Africa and the UAE are at the forefront of market development, with key players like Cepheid making strides in the region. The competitive landscape is characterized by a mix of local and international companies striving to enhance diagnostic capabilities. However, the market faces challenges such as limited resources and varying regulatory environments, which can hinder growth. The World Health Organization emphasizes the need for improved diagnostic capacity in the region to combat infectious diseases effectively.
Key Players and Competitive Insights
Key Companies in the Influenza Diagnostics Market include






Industry Developments
In the Influenza Diagnostics Market, recent developments indicate significant advancements in technology and increased strategic collaborations among key players. Companies such as Roche Diagnostics and Abbott Laboratories have recently enhanced their diagnostic capabilities with improved test accuracy and faster results, reflecting a growing demand for efficient influenza testing solutions. Thermo Fisher Scientific is expanding its portfolio, focusing on respiratory virus panels that include influenza tests, which is influencing market growth positively. Merger and acquisition activities are notable, with Siemens Healthineers acquiring certain assets from various health technology firms, strengthening their position in the diagnostics sector.
GenMark Diagnostics has seen growth within the market, primarily due to its rapid molecular diagnostic platforms that cater to influenza testing. Companies like bioMérieux and Quidel Corporation are also innovating their testing methodologies to adapt to seasonal flu variations and emerging strains, enhancing their competitive edge. The ongoing global health challenges create a heightened focus on respiratory illnesses, propelling growth and innovation across the influenza diagnostics landscape, thus affirming the importance of reliable and rapid testing solutions as a key priority for healthcare providers worldwide.
Future Outlook
Influenza Diagnostics Market Future Outlook
The Influenza Diagnostics Market is projected to grow at 8.33% CAGR from 2025 to 2035, driven by technological advancements, increasing awareness, and rising healthcare expenditures.
New opportunities lie in:
- Development of rapid point-of-care testing devices Integration of AI for predictive analytics in diagnostics Expansion of telehealth services for remote diagnostics
By 2035, the market is expected to achieve substantial growth, driven by innovation and increased demand. However, molecular diagnostics are emerging as the fastest-growing segment, driven by their higher sensitivity, accuracy, and ability to detect low viral loads, making them increasingly preferred for precise clinical decision-making.
Market Segmentation
Influenza Diagnostics Market End User Outlook
- Hospitals
- Diagnostic Laboratories
- Research Institutions
- Home Care Settings
Influenza Diagnostics Market Application Outlook
- Diagnostic Testing
- Surveillance
- Research and Development
- Public Health
- Clinical Trials
Influenza Diagnostics Market Product Type Outlook
- Rapid Diagnostic Tests
- Molecular Diagnostic Tests
- Serological Tests
- Viral Culture Tests
Report Scope
| MARKET SIZE 2024 | 9.26(USD Billion) |
| MARKET SIZE 2025 | 10.14(USD Billion) |
| MARKET SIZE 2035 | 22.43(USD Billion) |
| COMPOUND ANNUAL GROWTH RATE (CAGR) | 8.33% (2025 - 2035) |
| REPORT COVERAGE | Revenue Forecast, Competitive Landscape, Growth Factors, and Trends |
| BASE YEAR | 2024 |
| Market Forecast Period | 2025 - 2035 |
| Historical Data | 2019 - 2024 |
| Market Forecast Units | USD Billion |
| Key Companies Profiled | Roche Diagnostics (CH), Abbott Laboratories (US), Thermo Fisher Scientific (US), BD (Becton, Dickinson and Company) (US), Siemens Healthineers (DE), Cepheid (US), Hologic (US), Quidel Corporation (US) |
| Segments Covered | Application, Product Type, End User |
| Key Market Opportunities | Integration of rapid testing technologies enhances efficiency in the Influenza Diagnostics Market. |
| Key Market Dynamics | Technological advancements in rapid testing are reshaping the competitive landscape of the influenza diagnostics market. |
| Countries Covered | North America, Europe, APAC, South America, MEA |
Market Highlights
FAQs
What is the projected market valuation of the Influenza Diagnostics Market by 2035?
The Influenza Diagnostics Market is projected to reach a valuation of 22.43 USD Billion by 2035.
What was the market valuation of the Influenza Diagnostics Market in 2024?
In 2024, the market valuation of the Influenza Diagnostics Market was 9.26 USD Billion.
What is the expected CAGR for the Influenza Diagnostics Market during the forecast period 2025 - 2035?
The expected CAGR for the Influenza Diagnostics Market during the forecast period 2025 - 2035 is 8.33%.
Which companies are considered key players in the Influenza Diagnostics Market?
Key players in the Influenza Diagnostics Market include Roche, Abbott, Thermo Fisher Scientific, Siemens Healthineers, BD, Cepheid, Hologic, BioMérieux, and Quidel.
What are the main applications of influenza diagnostics and their market values?
The main applications include Diagnostic Testing valued at 8.5 USD Billion, Surveillance at 3.5 USD Billion, and Public Health at 4.5 USD Billion by 2035.
What types of tests are included in the Influenza Diagnostics Market?
The market includes Molecular Tests, Antigen Tests, Serological Tests, Rapid Diagnostic Tests, and Culture Tests, with projected values reaching 6.67 USD Billion and above by 2035.
How do end-user segments contribute to the Influenza Diagnostics Market?
End-user segments such as Hospitals and Diagnostic Laboratories are projected to reach 6.67 USD Billion and 5.05 USD Billion respectively by 2035.
What is the significance of research and development in the Influenza Diagnostics Market?
Research and Development is projected to grow from 1.0 USD Billion to 2.5 USD Billion by 2035, indicating its increasing importance in the market.
How does the market for rapid diagnostic tests compare to other test types?
Rapid Diagnostic Tests are expected to grow to 4.8 USD Billion by 2035, showcasing their competitive position among other test types.
What trends are influencing the growth of the Influenza Diagnostics Market?
Trends such as technological advancements and increased demand for rapid testing are likely to drive growth in the Influenza Diagnostics Market during the forecast period.
Research Approach
Secondary Research
The secondary research process involved comprehensive analysis of infectious disease surveillance databases, regulatory filings, peer-reviewed virology journals, clinical microbiology publications, and authoritative public health organizations. Key sources included the Centers for Disease Control and Prevention (CDC) including FluView surveillance data and Influenza Hospitalization Surveillance Network (FluSurv-NET), World Health Organization (WHO) Global Influenza Programme (GIP) and Global Influenza Surveillance and Response System (GISRS), European Centre for Disease Prevention and Control (ECDC), US Food & Drug Administration (FDA) in vitro diagnostic device clearances and Emergency Use Authorizations (EUAs), Biomedical Advanced Research and Development Authority (BARDA), National Institute of Allergy and Infectious Diseases (NIAID), National Institutes of Health (NIH), Health and Human Services (HHS) ASPR division, Public Health England (UK Health Security Agency), EU Eurostat Health Database, WHO FluNet, CDC Outpatient Influenza-like Illness Surveillance Network (ILINet), National Respiratory and Enteric Virus Surveillance System (NREVSS), and national health ministry surveillance reports from key markets.
These sources were employed to gather epidemiological statistics, regulatory approval data for RIDTs and molecular assays, clinical sensitivity/specificity studies, seasonal outbreak trends, and competitive landscape analysis for rapid antigen tests, RT-PCR platforms, loop-mediated isothermal amplification (LAMP), and emerging CRISPR-based diagnostics.
Primary Research
Qualitative and quantitative insights were obtained by interviewing supply-side and demand-side stakeholders during the primary research process. CEOs, VPs of Research & Development, regulatory affairs chiefs, and commercial directors from in vitro diagnostic (IVD) manufacturers, molecular diagnostic platform developers, and rapid test kit producers comprised supply-side sources. Demand-side sources included procurement leads from hospitals, independent diagnostic laboratories, urgent care centers, and academic medical institutions, as well as laboratory directors, infectious disease specialists, clinical microbiologists, and point-of-care testing coordinators. Primary research has confirmed product development pipelines for multiplex respiratory panels, validated market segmentation across test types (RIDT, molecular, viral culture), and gathered insights on laboratory adoption patterns, CLIA-waived testing expansion, pricing strategies for rapid vs. molecular assays, and reimbursement dynamics for influenza testing.
Primary Respondent Breakdown:
• By Designation: C-level Primaries (32%), Director Level (34%), Others (34%)
• By Region: North America (32%), Europe (30%), Asia-Pacific (28%), Rest of World (10%)
Market Size Estimation
Revenue mapping and testing volume analysis were employed to determine the global market valuation. The methodology comprised the following:
• The identification of over 50 main diagnostic manufacturers in North America, Europe, Asia-Pacific, and Latin America
• Product mapping between rapid influenza diagnostic tests (RIDTs), RT-PCR assays, isothermal amplification tests, viral culture media, and emerging CRISPR-based diagnostic platforms
• Examination of influenza diagnostic portfolios and respiratory panel assays' annual revenues, as reported and modeled
• In 2024, the coverage of manufacturers will account for 75-80% of the global market share.
• Segment-specific valuations across hospital laboratories, point-of-care settings, and home-based testing environments are derived through extrapolation using bottom-up (test volume × ASP by country/setting) and top-down (manufacturer revenue validation) approaches.
Kindly complete the form below to receive a free sample of this Report
Customer Stories

“This is really good guys. Excellent work on a tight deadline. I will continue to use you going forward and recommend you to others. Nice job”

“Thanks. It’s been a pleasure working with you, please use me as reference with any other Intel employees.”

“Thanks for sending the report it gives us a good global view of the Betaïne market.”

“Thank you, this will be very helpful for OQS.”

“We found the report very insightful! we found your research firm very helpful. I'm sending this email to secure our future business.”

“I am very pleased with how market segments have been defined in a relevant way for my purposes (such as "Portable Freezers & refrigerators" and "last-mile"). In general the report is well structured. Thanks very much for your efforts.”

“I have been reading the first document or the study, ,the Global HVAC and FP market report 2021 till 2026. Must say, good info! I have not gone in depth at all parts, but got a good indication of the data inside!”

“We got the report in time, we really thank you for your support in this process. I also thank to all of your team as they did a great job.”




