Top Industry Leaders in the Influenza Diagnostics Market
 Influenza Diagnostics Companies Key Companies" width="600" height="300">Disclaimer: List of key companies in no particular orderLatest Influenza Diagnostics Companies Update
Influenza Diagnostics Companies Key Companies" width="600" height="300">Disclaimer: List of key companies in no particular orderLatest Influenza Diagnostics Companies Update
-
March 2023: Lucira Health, Inc., a medical technology company, made two major announcements: (1) the nationwide release of its innovative Lucira COVID-19 & Flu Home Test in the United States; and (2) the inclusion of the combination test in the Australian Register of Therapeutic Goods (ARTG) for use by healthcare professionals at the point-of-care. Emergency use authorization (EUA) for OTC use at home and other non-laboratory settings was given to the COVID-19 & Flu Home Test by the U.S. Food and Drug Administration (FDA). In clinical studies, the Lucira COVID-19 & Flu Home Test, a molecular test, performed comparably to extremely sensitive lab-based PCR tests for both COVID-19 and Influenza. The simple, combined test just requires a superficial nose swab and provides results in 30 minutes or less.
-
April 2023: The Truenat H3N2/H1N1 for H3N2 and H1N1 differential diagnostic was released by India's Molbio Diagnostics in April 2023. The test has been sanctioned by CDSCO and turnaround time from sample to result is less than an hour. In order to confirm the diagnosis of Influenza infections, the first point-of-care Real-Time PCR test, Truenat H3N2/H1N1, has been developed. Over 3500 primary and community healthcare centers and over 1200 private laboratories and hospitals in India use Truenat equipment, so patients may get a rapid diagnosis at any point in the healthcare delivery chain. An increase in H3N2 and H1N1 influenza infections has been reported in India recently. The airborne viruses can continue to spread from person to person, which might lead to a global influenza pandemic.
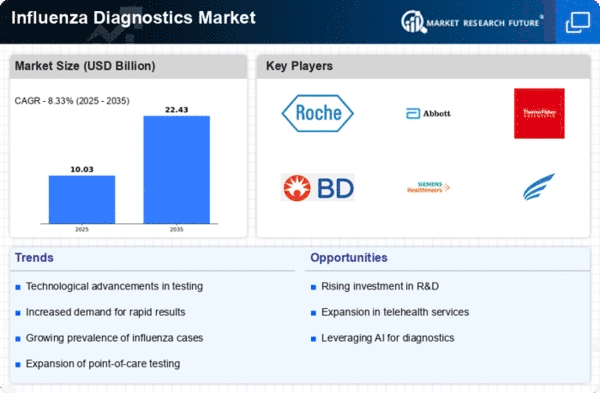
List of Influenza diagnostics Key companies in the market
- Coris BioConcept
- DiaSorin S.p.A
- Alere Inc.
- Lonza
- Ortho-Clinical Diagnostics
- Qiagen
- Wako
- Abbott
- Analytik Jena AG
- Becton, Dickinson and Company,
- Biocartis,
- bioMérieux Inc,
- Cepheid,
- DiaSorin S.p.A., F.
- Hoffmann-La Roche Ltd