Choroidal Neovascularization Market
ID: MRFR/HC/25867-HCR
100 Pages
Rahul Gotadki
Last Updated: April 06, 2026
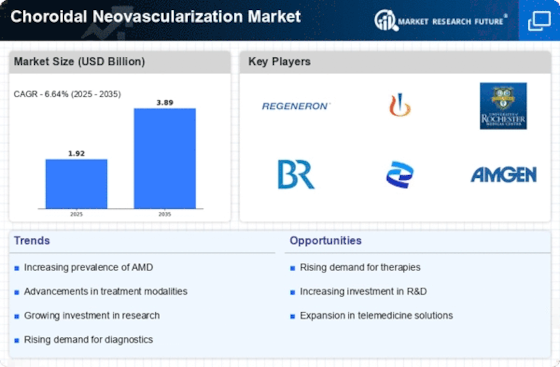
Choroidal Neovascularization Market Research Report By Treatment Type (Anti-Angiogenic Therapy, Laser Photocoagulation, Photodynamic Therapy, Verteporfin Sodium, Ranibizumab, Bevacizumab, Aflibercept, Pegaptanib Sodium), By Indication (Age-Related Macular Degeneration (AMD), Diabetic Macular Edema (DME), Central Serous Chorioretinopathy (CSC), Polypoidal Choroidal Vasculopathy (PCV), Myopic Macular Degeneration (MMD), Other Indications), By Route of Administration (Intravitreal, Subconjunctival, Periocular, Oral), By End User (Hospitals, Ophthalmology Clinics, Ambulatory Surgical Centers, Research Centers) and By Regional (North America, Europe, South America, Asia Pacific, Middle East and Africa) - Growth & Industry Forecast 2025 To 2035
.png)