North America: Expanding patient population receiving treatment
North America is the most regulation-driven and quality-sensitive region for thyroid active pharmaceutical ingredients (APIs) as it has a large patient population receiving treatment and a mature diagnostic infrastructure for the treatment of thyroid diseases and stringent requirements for the performance of narrow therapeutic index (NTI) products. Demand in North America is primarily driven by the chronic management of hypothyroidism, but API suppliers face a number of challenges in meeting increasing compliance requirements, particularly with regard to maintaining potency, controlling impurities, and managing lifecycle changes. On the demand side of the equation, the United States has a significant prevalence of thyroid disease that is well documented. Based on the U.S. National Institute of Diabetes, Digestive, and kidney diseases (NIDDK), almost five out of every 100 Americans aged 12 and above are diagnosed with hypothyroidism. Many of these individuals have mild or subtle hypothyroidism, leading to a large and ongoing pool of individuals who will need therapy throughout their lifetime.
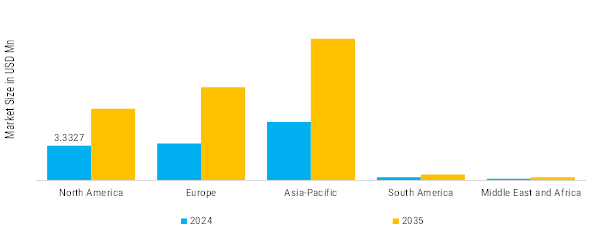
Europe: Expanding thyroid APIs
The Thyroid API Market has been classified as a highly regulated, epidemiologically significant, quality-driven area of the market in Europe. Demand for thyroid APIs is significantly affected by the high incidence of diagnosed or undiagnosed thyroid diseases, healthcare systems offering either full or partial coverage for all citizens in most European nations, and the presence of a uniform level of regulatory oversight through agencies such as the EMA (European Medicines Agency) and EDQM (European Directorate for the Quality of Medicines). Europe has a stronger emphasis on the consistency of therapies, interchangeability and the long-term safety of patients, which has an effect on establishing API quality threshold standards and supplier qualifications when compared to other price sensitive markets.
Asia Pacific: Fastest Growing thyroid APIs
The Asia-Pacific (APAC) region is the fastest growing market and structurally most diverse in the Thyroid API landscape. It is characterized by a significant number of populations and their geographically dispersed nature, as well as mixed levels of diagnostic penetration and the wide range in levels of iodine nutrition, together with the rapid increase in pharmaceutical manufacturing capacity in this region. Unlike North America and Europe, where the principal driver of growth is a fully established treatment ecosystem, APAC has both significant latent Epidemiological need for treatment and a very strong supply of APIs for the treatment of thyroid disease so it acts as both a driver of growth for thyroid API consumption AND a critical global manufacturer of APIs for thyroid disease treatments In terms of epidemiology, the APAC region carries a disproportionately high burden of the global thyroid disease burden due to the sheer scale of its populations. Public health assessments conducted on a regional basis have shown that millions of people in China, India, Japan, and Southeast Asia have thyroid disorders with most of them classified as having hypothyroidism.
South America: Growing thyroid APIs
South America is characterized by relatively moderate penetration, yet, at the same time, growth rates for Thyroid API in South America are rapidly changing due to the combination of high prevalence of thyroid disease, a rapid increase in the number of diagnostic assays and a reliance on imported APIs. In addition, South America’s market environment is defined by a high degree of reliance on public healthcare systems, variances & fluctuations in South American economies and an emerging trend towards access to essential medicines, rather than towards premium pricing models. Furthermore, a core component of the Thyroid Therapeutics category is a key inclusion within the Essential Medicines programme in virtually all of the largest economies in South America. In addition to the economic aspects of Hypothyroidism, the region has a significant epidemiological burden associated with thyroid disorders due to a combination of the high prevalence of autoimmune thyroid diseases, fluctuations in historical iodine levels and population demographic ageing.
Middle East & Africa: Emerging thyroid APIs
The MEA (Middle East and Africa) region is a structurally different and underdeveloped market for thyroid active pharmaceutical ingredients (APIs), where healthcare access is inconsistent; the iodine nutrition has not improved; the escalating number of people with non-communicable diseases (NCDs); and a high dependence on imported pharmaceutical products to treat thyroid disorders is generally the case across the region. The MEA market is therefore driven by access to therapies, public health initiatives, and the gradual expansion of diagnostic capabilities rather than by market efficiency in optimizing therapies. Consequently, the rate of demand for thyroid API in the MEA is expected to remain low, but will eventually show positive structural growth over time, as more people are treated with these medicines.













