Dried Blood Spot Testing Market Summary
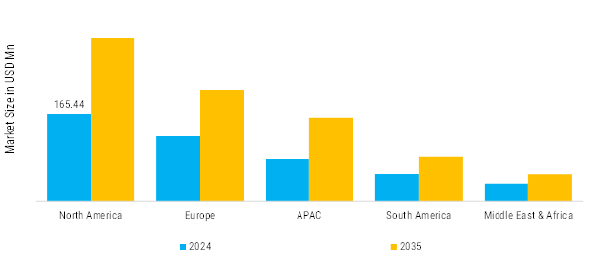
As per Market Research Future analysis, Dried Blood Spot Testing Market Size was valued at USD 451.57 Million in 2024. The Dried Blood Spot Testing Industry is projected to grow from USD 476.03 Million in 2025 to USD 810.51 Million by 2035, exhibiting a compound annual growth rate (CAGR) of 4.92% during the forecast period 2025 - 2035. North America led the market with over 36.64% share, generating around USD 165.44 Million in revenue.
The Dried Blood Spot Testing Market is driven by increasing demand for minimally invasive diagnostic techniques, expansion of newborn screening programs, and rising adoption of decentralized testing, enabling efficient sample collection, transport, and large-scale disease detection across diverse healthcare settings globally.
According to UNICEF, over 140 million newborns are screened annually worldwide, supporting early disease detection, while the World Health Organization (WHO) highlights growing demand for accessible diagnostics, reinforcing adoption of dried blood spot testing for large-scale and remote healthcare applications.
Key Market Trends & Highlights
The Dried Blood Spot Testing Market is being reshaped by a cluster of interconnected trends that span technology, regulation, business models, and healthcare‑system priorities.
- Newborn screening dominates with 49% share, driven by increasing global focus on early disease detection programs.
- Elution-based testing leads with 58% share, supported by high accuracy and widespread laboratory adoption globally.
- North America leads with over 36.64% share in 2024, driven by strong newborn screening coverage and advanced diagnostic infrastructure.
- Europe market valued at USD 135.47 million in 2024, accounting for 30% share, supported by preventive healthcare policies.
Market Size & Forecast
| 2024 Market Size | 451.57 (USD Million) |
| 2035 Market Size | 810.51 (USD Million) |
| CAGR (2025 - 2035) | 4.92% |
Major Players
Companies such as Ahlstrom Oyj, Revvity Inc, Archimedlife Gmbh, Qiagen, Eastern Business Forms, Inc., Rda Spot Inc., Salimetrics, Llc., Cytiva, Centogene Gmbh, Neoteryx, LLC. are some of the major participants in the global market.