Market Analysis
In-depth Analysis of Biosimilars Market Industry Landscape
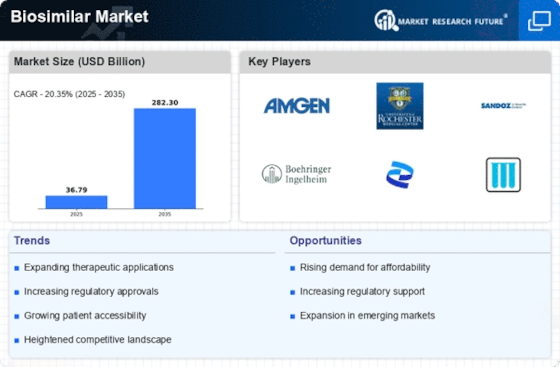
Biosimilars, which comprise of biologic drugs that don't show clinically huge varieties, are dependent upon the impact of monetary variables, administrative systems, and medical services requests. The biosimilars market is encountering development as the need might arise for practical biologic treatments; thusly, they have turned into a fundamental component of medical services availability.
Open doors for Market Passage and Patent Termination: The lapse of originator biologic licenses gives biosimilar makers possibilities to infiltrate the market. Biosimilars can possibly present contest as selectiveness periods close, consequently introducing more reasonable other options and cultivating a more serious climate. The administrative system and endorsement processes are critical variables that impact the elements of the biosimilars market. The timing and progress of the market section are influenced by thorough administrative prerequisites and endorsement processes, which ensure that biosimilars stick to raised measures for wellbeing, viability, and quality. The biosimilars market displays an extensive variety of treatment choices because of the speculation of organizations in innovative work for various restorative spaces. As an outcome, the market is dynamic and continually developing. The reception of biosimilars by doctors and patients significantly affects market elements. Mindfulness and schooling efforts add to the advancement of confidence in the wellbeing and adequacy of these items. Biosimilar makers experience imposing contests as they endeavor to get a piece of the market. To accomplish this, they utilize creative promoting strategies, cutthroat evaluating structures, and the foundation of notorieties for quality and trustworthiness. The biosimilars market is encountering development in arising economies as a way to determine unfulfilled clinical prerequisites and improve openness and moderateness to overcome monetary impediments. Biosimilar Lifecycle The board: Market elements are affected by lifecycle the executives’ techniques, which incorporate the improvement of biosimilar mixes and the presentation of biosimilars of the future. Makers try to grow their scope of items and support an upper hand throughout the span of the item's life cycle. Market elements are fundamentally impacted by essential coordinated efforts and associations that happen among drug organizations, biosimilar producers, and administrative bodies. The essential target of these unions is to advance improvement processes, smooth out specific information, and more prominent proficiently explore mind boggling administrative conditions. The use of biosimilars in the administration of persistent illnesses and oncology addresses a huge improvement that is influencing the elements of the market. Biosimilars give monetarily reasonable substitutes to fundamental medicines, in this manner working with an essential change in the way to deal with overseeing such circumstances. Evaluating Tensions and Worldwide Financial Elements: Valuing pressures and monetary variables impact the elements of the biosimilars market. The fragile harmony among moderateness and innovative work costs presents obstructions, convincing makers to change their evaluating techniques as per the financial matters of medical care.



















Leave a Comment