Biosimilars Market Summary
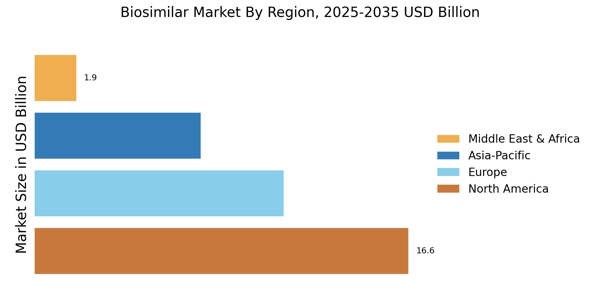
As per Market Research Future analysis, the Biosimilars Market Size was estimated at 36.79 USD Billion in 2024. The Biosimilar industry is projected to grow from 44.28 USD Billion in 2025 to 282.3 USD Billion by 2035, exhibiting a compound annual growth rate (CAGR) of 20.35% during the forecast period 2025 - 2035.
The Biosimilars Market is primarily driven by rising demand for cost-effective biologic alternatives and increasing healthcare expenditure, supported by patent expirations of major biologics and growing focus on improving treatment accessibility, enabling wider patient reach and sustainable healthcare systems globally.
According to the World Health Organization (WHO), biological therapies are essential for treating chronic diseases, while the Institute for Health Metrics and Evaluation (IHME) highlights increasing burden of cancer and autoimmune diseases, significantly driving demand for affordable biosimilar treatments worldwide.
Key Market Trends & Highlights
The Biosimilars Market is poised for substantial growth driven by increasing demand for cost-effective treatments and supportive regulatory frameworks.
- Increasing burden of chronic diseases globally is significantly driving demand for cost-effective biosimilar therapies and biologic alternatives.
- Oncology segment leads with 63% share, supported by rising global cancer prevalence and treatment demand.
- Monoclonal antibodies dominate with 59% share, reflecting strong adoption in targeted therapies.
- Hospital pharmacies hold 62% share, ensuring effective distribution of complex biologic treatments across healthcare systems.
Market Size & Forecast
| 2024 Market Size | 36.79 (USD Billion) |
| 2035 Market Size | 282.3 (USD Billion) |
| CAGR (2025 - 2035) | 20.35% |
Major Players
Companies such as Amgen (US), Roche (CH), Sandoz(CH), Boehringer Ingelheim (DE), Pfizer (US), Mylan (US), Samsung Bioepis (KR), Teva (IL), Celltrion (KR) are some of the major participants in the global market.