Bioengineered Protein Drugs Market
ID: MRFR/Pharma/7520-HCR
100 Pages
Satyendra Maurya
Last Updated: April 06, 2026
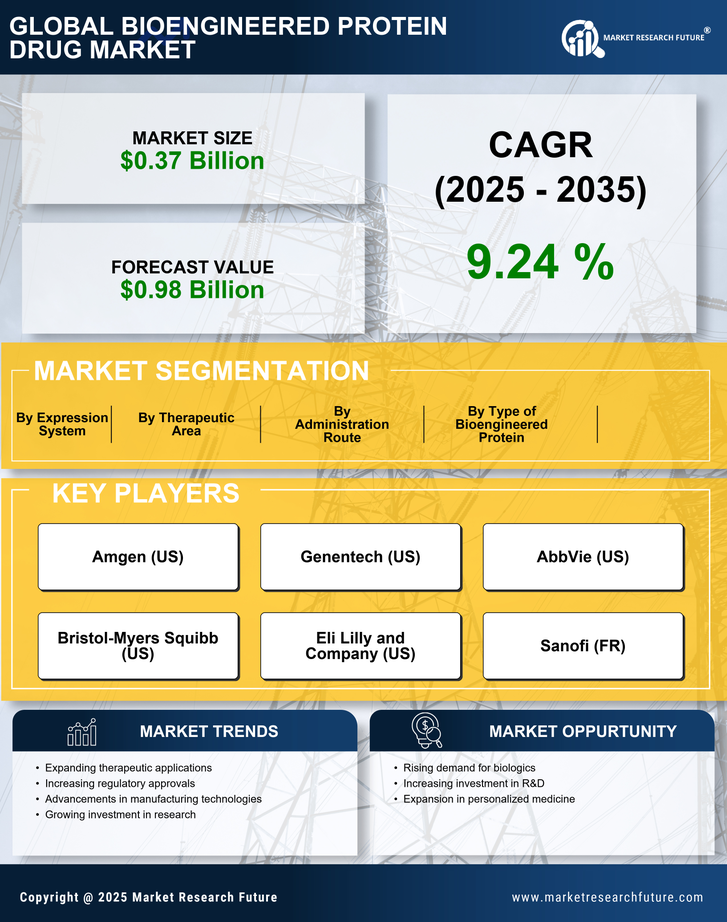
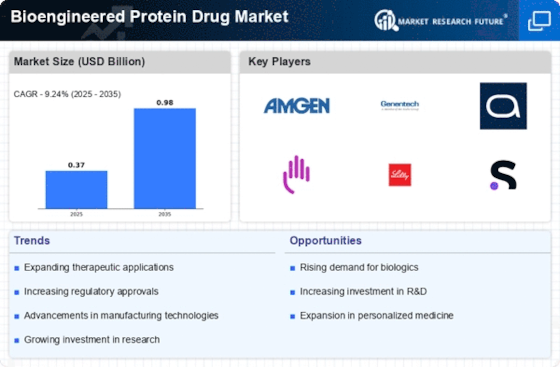
Bioengineered Protein Drug Market Research Report By Therapeutic Area (Oncology, Immunology, Metabolic Disorders, Neurological Disorders, Cardiovascular Diseases), By Type of Bioengineered Protein (Monoclonal Antibodies, Fusion Proteins, Enzymes, Hormones, Growth Factors), By Administration Route (Intravenous, Subcutaneous, Intramuscular, Intraperitoneal, Topical), By Expression System (Mammalian Cell Culture, Bacterial Expression, Yeast Expression, Plant Expression, Insect Cell Expression) and By Region - Growth & Industry Forecast 2025 To 2035