ID: MRFR/HC/41502-HCR
200 Pages
Rahul Gotadki
Last Updated: April 13, 2026
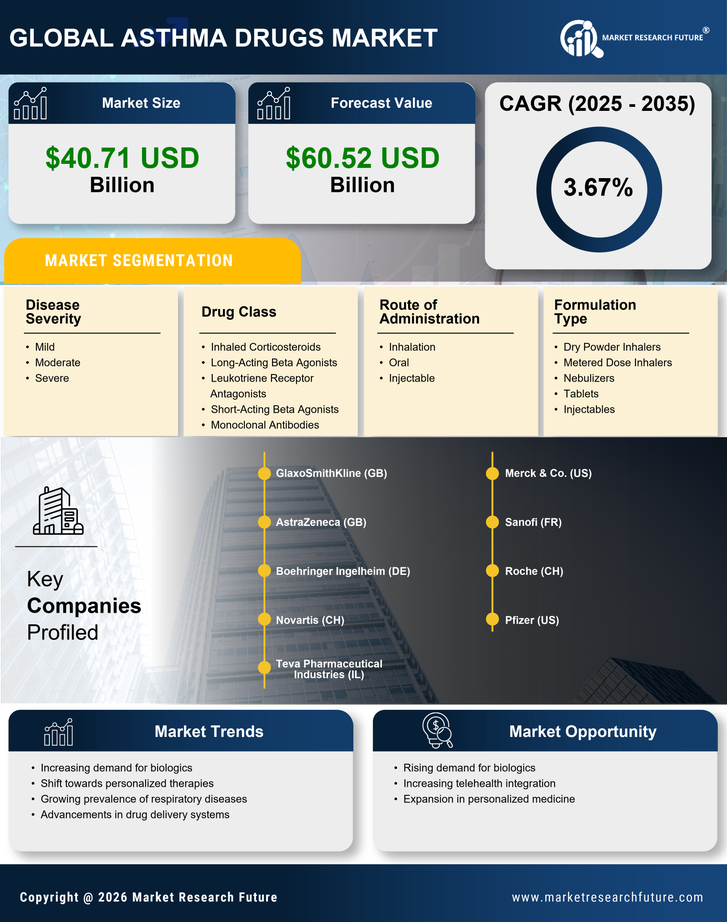
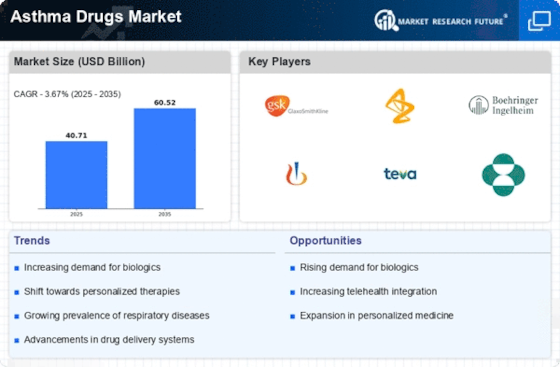
Asthma Drugs Market Research Report: Size, Share, Trend Analysis By Drug Class (Inhaled Corticosteroids, Long-Acting Beta Agonists, Leukotriene Receptor Antagonists, Short-Acting Beta Agonists, Monoclonal Antibodies), By Route of Administration (Inhalation, Oral, Injectable), By Disease Severity (Mild, Moderate, Severe), By Formulation Type (Dry Powder Inhalers, Metered Dose Inhalers, Nebulizers, Tablets, Injectables) andBy Regional (North America, Europe, South America, Asia Pacific, Middle East and Africa) - Growth Outlook & Industry Forecast 2025 To 2035