As part of the initial research process, stakeholders from both the supply and demand sides were interviewed to get both qualitative and quantitative information. CEOs, VPs of Product Development, heads of regulatory affairs, R&D directors, and marketing directors from medical adhesive manufacturers, medical device OEMs, and raw material suppliers were some of the supply-side sources. Chief surgeons, operating room directors, procurement managers from hospitals and ambulatory surgical centers, wound care experts, dentists, and medical device assembly engineers were some of the demand-side sources. Primary research confirmed market segmentation across resin types and application areas. It also confirmed product pipeline timelines for next-generation biocompatible adhesives and gathered information on how surgical adhesives are used, how they are priced, how reimbursement works for tissue adhesives, and the rules that must be followed by the industry.
Primary Respondent Breakdown:
By Designation: C-level Primaries (32%), Director Level (31%), Others (37%)
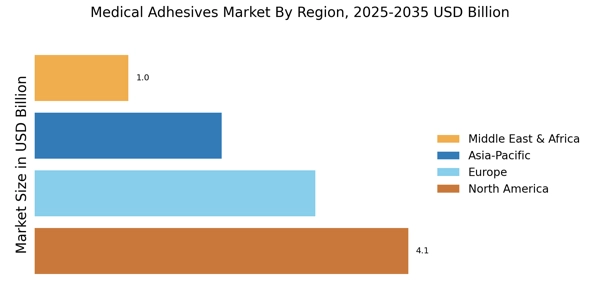
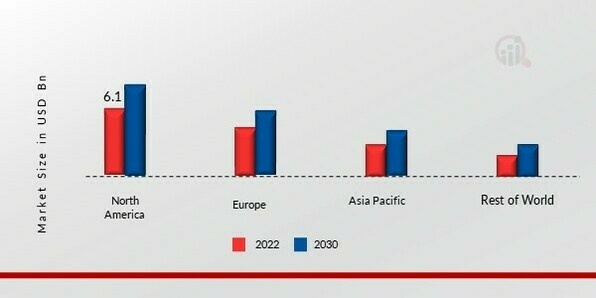
By Region: North America (32%), Europe (30%), Asia-Pacific (28%), Rest of World (10%)
Global market valuation was derived through revenue mapping and procedure volume analysis. The methodology included:
Identification of 50+ key manufacturers across North America and Europe



 出典:二次研究、一次研究、市場調査未来データベース、およびアナリストレビュー
出典:二次研究、一次研究、市場調査未来データベース、およびアナリストレビュー