真空采血管市场概览
p2023 年真空采血管市场规模价值 84.1 亿美元。真空采血管市场行业预计将从 2024 年的 89.5 亿美元增长到 2032 年的 139.2 亿美元,在预测时间范围内(2024 - 2032 年)的复合年增长率 (CAGR) 为 5.67%。由于输血量的增加,采血设备变得越来越流行,而增加对真空采血设备的支出是推动市场增长和收入的主要市场驱动力。
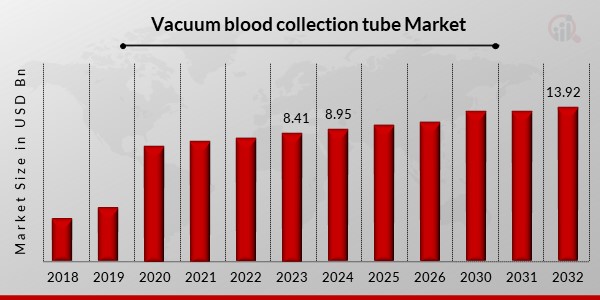
来源:二手资料研究、一手资料研究、MRFR 数据库和分析师评论
真空采血管市场趋势
ul
p真空采血管市场的复合年增长率是由真空采血管设备支出的增加推动的。医疗机构输血量的增加、标本检测的频繁进行以及创新采血设备的兴起正在推动市场的发展。在手术和灾难情况下输血的使用增加推动了该行业的发展。随着心脏病、肝病、肾病、肺病和其他慢性疾病的发病率上升,每年进行的手术数量也在增加。
此外,需要利用血液样本进行血液检测以区分疾病。真空采血装置用于收集和储存血液进行分析。该行业的发展取决于血液保存的需求和选择。由于政府鼓励卫生和医疗设施的举措,最终用户对真空采血装置的兴趣有所扩大。政府运营和医院正在推动对真空采血管的需求,预计这将提振市场。此外,根据美国国家医学图书馆 2022 年 4 月发布的研究,血液中心应集中精力制定举措和法规,以加强其国家的血液供应,因为该病毒不会对血液安全构成直接威胁。诸如推出家用采血管等新产品之类的举措可能会在预计的时间内推动市场增长。
COVID-19 损害了多个商业领域;另一方面,真空采血管市场销售额有所增长。预计大流行的出现将导致需求增加。由于疫情,对真空采血管的需求自然增加。由于大规模的检测工作,该行业在大流行期间经历了破纪录的需求,这意味着该行业的收入和利润有所增加。事实上,在可预见的未来,需求将保持高位。
例如,根据世界卫生组织 (WHO) 的数据,癌症是 2020 年全球最常见的死亡原因,造成 1000 万人死亡。此外,这些测试通常在手术前进行,用于治疗慢性水肿、血栓、莫名其妙的不适和僵硬等情况。此外,它们还能提高人们对血友病 A 和 B 等遗传疾病影响的认识。这些变量可能会帮助这个市场赚钱。因此,由于真空采血设备支出的增加,预计整个预测期内对真空采血的需求将会增加。从而推动了真空采血管市场收入的增长。
真空采血管市场细分洞察
h3
真空采血管类型洞察 p真空采血管市场根据类型细分,包括凝胶和凝块激活剂管、肝素管和血清分离管。血清分离管占据了市场主导地位,2022 年占市场收入的 37%(29.2 亿美元)。这是由于分离管具有诸多优势,例如可以将血清与血液中的细胞成分彻底分离,无需分离血清,可以减少危险物质的气溶胶化,从而缩短分析前的重组时间,并且运输方便。
真空采血管应用洞察
p真空采血管市场根据应用可细分为血常规检查、生化测试和
凝血测试。血常规检查类别在 2022 年创造了最多的收入,约占 42%(33 亿美元),因为其在疾病诊断中的用途十分广泛。定期血液检测可以评估循环系统中细胞、蛋白质分子和其他成分的数量。此外,这些检测结果可能在数小时内即可获得,以便在极端情况下及时治疗。预计在预测期内,凝血检测将显著扩展。
图 1:真空采血管市场(按应用划分),2022 年及以后2032 年(十亿美元)
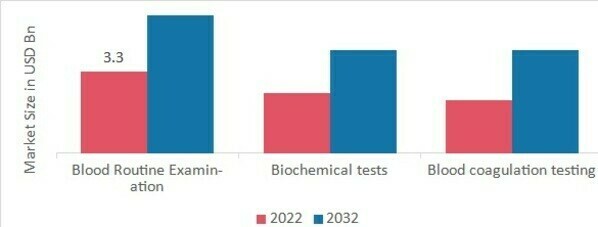
来源:二手研究、一手研究、MRFR 数据库和分析师评论
真空采血管最终用户洞察
p 真空采血管市场细分基于最终用户,包括医院、诊所和病理实验室。病理实验室占据了市场主导地位,2022 年占市场收入的 49%(38.7 亿美元),这是因为许多疾病的发病率很高,包括
乙肝病毒、艾滋病毒、艾滋病、癌症、糖尿病和心血管疾病,这些疾病需要在病理实验室进行各种血液检测来确定疾病的严重程度。
真空采血管区域洞察
p按地区划分,该研究提供了北美、欧洲、亚太地区和世界其他地区的市场洞察。得益于先进的医疗基础设施、知名的真空采血管供应商以及政府的大力支持,预计北美真空采血管市场将在预测期内快速增长和扩张。此外,不断增加的现有医疗机构数量将推动该地区的市场增长。
此外,市场报告中研究的主要国家是美国、加拿大、德国、法国、英国、意大利、西班牙、中国、日本、印度、澳大利亚、韩国和巴西。
图 2:2022 年各地区真空采血管市场份额(十亿美元)
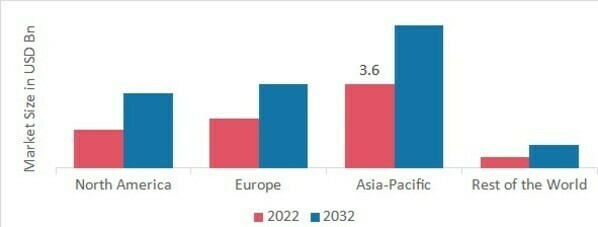
来源:二手研究、一手研究、MRFR 数据库和分析师评论
欧洲地区的真空采血管市场占由于公众和私人对献血和安全转移流程的认识不断提高,加上有利的官方和企业活动将在预测的时间范围内推动市场增长,因此市场份额排名第二。此外,德国真空采血管市场占有最大的市场份额,而英国真空采血管市场将在预测的时间范围内在欧洲地区稳步扩张。
在预测的时间范围内,亚太真空采血管市场将主导这一市场。这是由于人口增长和疾病流行病蔓延。此外,包括癌症和心脏病在内的慢性病发病率增加也促进了亚太地区的崛起。此外,中国的真空采血管市场占据市场份额的主导地位,预计印度真空采血管市场将在亚太地区以快速的复合年增长率大幅扩张。
真空采血管主要市场参与者和竞争洞察
p领先的市场参与者在研发方面投入巨资,以扩大其生产部门和产品线,这将有助于真空采血管市场在全球范围内的增长。市场参与者也在采取各种有机或无机战略方法来加强和扩大其影响力,重要的市场发展包括新产品组合、合同交易、并购、资本支出、增加投资以及与其他组织的战略联盟。企业也在制定营销策略,例如数字营销、社交媒体影响力和内容营销,以扩大其利润空间。真空采血管行业必须提供具有成本效益且可持续的选择,才能在分散且充满活力的市场环境中生存。
在本地生产以最大限度地降低运营成本并为客户提供售后服务是真空采血管行业各组织用来使客户受益并获取尚未开发的市场份额和收入的关键商业策略之一。真空采血管行业最近为医药和服务行业带来了显著的优势。此外,越来越多的行业参与者正在利用和采用这些技术,尖端技术也得到了大幅发展。真空采血管市场的主要参与者,包括 Becton、Dickinson and Company、Sunphoria Co. Ltd.、Narang Medical Limited、CML Biotech、Bio – X、Labtech Disposables、河北新乐科技股份有限公司、Greiner Bio-One International、Biosigma、Medtronic plc. 和 Terumo,正试图通过投资研发业务来扩大市场份额和需求,以提供可持续且经济实惠的解决方案。
Immunexpress Pty Ltd 是一家分子诊断公司,致力于开发用于治疗免疫疾病的临床检测方法。它通过量化免疫系统对病原体的反应来识别病因感染,从而推动败血症检测的变革。他们宣布,SeptiCyte RAPID EDTA血液兼容注射器将于2022年8月登陆欧洲市场。在医院,EDTA采血管用于血液学检测。
Abacus Dx成立于1966年,为医疗保健行业提供诊断病理学、临床研究和检测仪器。其客户包括商业和政府实验室、献血机构以及学术研究单位。他们于2022年2月推出了RNA Complete BCT,这是Streck备受赞誉的采血管系列的最新成员。
真空采血管市场的主要公司包括
ul
- Becton Dickinson and Company
h2
真空采血管行业发展 p
2022年8月:Immunexpress, Pty Ltd.宣布,SeptiCyte RAPID EDTA血液兼容注射器将于2022年8月登陆欧洲市场。在医院,EDTA采血管用于血液学检测。2022年5月:Tethis SpA推出了See.d,这是首个用于液体活检分析的血液样本。这项尖端技术完全自动化和标准化了血液样本采集过程中的设置。细胞部分在获得专利的纳米涂层SBS载玻片上逐渐稳定,以识别罕见细胞,而血浆则可用于无细胞成分分析。 See.d 对采集后 4-6 小时内在 EDTA 管中采集的新血液样本进行分析,以确保最佳的样本完整性。
2022 年 2 月:Abacus dx 于 2022 年 2 月推出了 RNA Complete BCT,这是 Streck 著名采血管系列的最新成员。
真空采血管市场细分
h3
真空采血管类型展望 ul
h3
真空采血管应用展望 ul
h3
真空采血管最终用户展望 ul
h3
真空采血管区域展望 p
北部美洲 p
欧洲 p
亚太地区 p
世界其他地区
















