全球囊性纤维化市场概览
p囊性纤维化市场规模在2023年的价值为60.1亿美元,预计将从2024年的68.4亿美元增长到2032年的204.5亿美元,预测期内(2024-2032年)的复合年增长率(CAGR)为12.95%。推动囊性纤维化扩张的一些关键市场驱动因素包括人们对这种渐进性遗传疾病的认识不断提高以及药物治疗的应用不断扩大。
2024年5月,Vertex Pharmaceuticals 的成功很大程度上依赖于特定的产品或服务,在过去几年中,该公司表现出了扩大和改变收入来源的强烈愿望。随着众多患者开始接受Casgevy(该公司与CRISPR技术合作开发的基因编辑细胞疗法)的治疗,Vertex制药公司正朝着实现其目标迈出重要一步。Casgevy于12月获得镰状细胞病(SCD)的批准,并于次月获得β地中海贫血的批准。Vertex制药公司目前在启动授权治疗中心、与医生和患者沟通以及与付款方洽谈方面取得了重大进展。除了启动治疗中心外,Vertex制药公司还在第一季度财报电话会议上讨论了细胞采集这一重要的启动环节。截至4月中旬,已从Casgevy目前获批的三个地区(美国、欧洲和中东)的五名患者身上采集了细胞样本。Arbuckle称赞这一里程碑是一项非凡的成就,考虑到Casgevy自获批以来的相对较短的时间,以及患者接受Casgevy治疗的复杂而漫长的过程。根据Vertex的统计数据,其药物Casgevy在FDA批准方面的排名高于其竞争对手蓝鸟生物的Lyfgenia。这两种药物于去年12月同时获得FDA批准。Bluebird公司透露,其SCD基因治疗药物仅从一名患者身上采集了细胞。
2024年1月,总部位于美国的Clarametyx Biosciences公司将开始招募Ib/IIa期临床试验的参与者。该试验旨在研究CMTX-101作为囊性纤维化(CF)患者辅助治疗的效果。这项Ib/II期临床试验(NCT06159725)由囊性纤维化基金会资助,旨在评估CMTX-101在美国稳定性囊性纤维化和慢性感染患者中的安全性。临床试验参与者将接受CMTX-101联合妥布霉素治疗,为期28天。CMTX-101是一种高度精准的抗生物膜药物,可提高抗生素的敏感性,从而可能降低抗生素耐药率。该候选药物具有拮抗DNABII蛋白的活性,可快速破坏生物膜并释放驻留细菌。这有助于通过人体免疫系统或抗生素治疗清除这些细菌。Ib期研究将有六名患者参与,剂量将逐渐增加。该试验将采用开放标签,这意味着患者和研究人员都将了解正在接受哪种治疗。剂量将包括5毫克、15毫克和30毫克。Richards表示,Ib期研究组的注册流程已经开始。
囊性纤维化市场趋势
ul
p在世界各地,囊性纤维化(CF)的患病率正在迅速超过其他遗传性疾病。根据囊性纤维化患者登记处的数据,目前美国有超过 30,000 人患有囊性纤维化,预计全球有 70,000 人患有这种疾病。还提到,每年约有 1,000 个新病例被诊断出来。因此,预计在整个预测期内,由于 CF 患者中呼吸系统疾病、胃肠道疾病和生殖系统疾病的患病率上升,对该疾病的治疗需求将会增加。采用新的治疗方法进行早期发现也增加了患者基础,从而改善了治疗,预计这将影响整个预期期内的市场增长。因此,这个因素正在推动市场复合年增长率。p
此外,囊性纤维化患者在世界范围内变得越来越普遍,这激发了该行业的发展。囊性纤维化的药物治疗在管理这种疾病方面取得了许多进展,有助于市场增长。该行业的崛起也受到目前用于治疗该疾病的市场趋势的推动。这是因为越来越多的候选治疗药物正在由大型企业生产,并且目前正在研发中。因此,制药公司正被迫开发针对CFTR蛋白异常的新型疗法。此外,Lumacaftor/Ivacaftor(一种可改善该疾病患者生活质量的增效剂)的上市也有助于市场的增长。
然而,由于即将推出的CF治疗药物,市场正在不断扩大。在预期的时期内,针对患者细胞缺陷的药物的推出将有助于市场扩张。市场扩张受到这种药物可能改变许多人生活的影响。市场也被认为主要受到该疾病患病率上升的推动。这反过来又加速了新开发的、用于危重患者护理的在研药物的推出。此外,许多潜在候选药物正处于开发的最后阶段,预计市场将很快复苏。例如,Vertex Pharmaceuticals Incorporated 最近报告了其用于治疗粘液纤毛清除的吸入甘露醇 III 期临床研究的令人鼓舞的结果。由于用于治疗各种严重疾病(例如粘液稀释剂、气道清除程序等)的技术先进的设备,市场预计也将增长。因此,预计这方面将加速全球囊性痤疮市场收入的增长。
囊性纤维化市场细分洞察:
h3
囊性纤维化治疗方法洞察 p囊性纤维化市场细分基于治疗方法,包括药物和设备。 2022 年囊性纤维化市场数据中,药物领域占据了绝大部分份额。公众对囊性纤维化潜在危险的认识不断提高、药物疗法使用率的上升以及全球范围内该疾病患病率的上升,是推动该类别扩张的一些关键驱动因素。
图 2:囊性纤维化市场(按治疗方法划分),2023 年及以后2032 年(十亿美元)
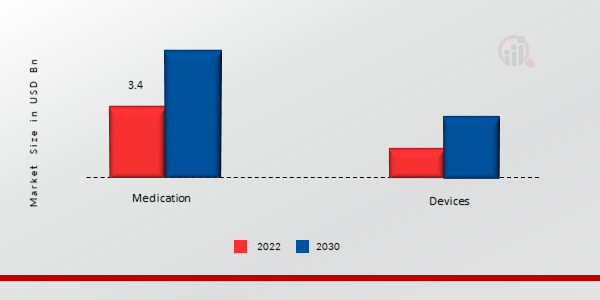
来源:二手研究、一手研究、MRFR 数据库和分析师评论
囊性纤维化给药途径洞察
p根据给药途径,囊性纤维化市场细分包括口服和吸入。口服药物在 2022 年占据了市场增长的主导地位,预计在 2024-2032 年预测期内,由于服用药物的简单便捷,口服药物将成为增长最快的细分市场。然而,如果出现易于给药且无侵入性操作风险的新药物制剂,口服药物制剂的市场份额可能会被吸入药物等其他剂型所取代。
囊性纤维化药物类别洞察
p囊性纤维化市场数据基于药物类别,包括胰酶补充剂、粘液溶解剂、支气管扩张剂、抗生素和 CFTR 调节剂。CFTR 调节剂细分市场在 2022 年占据了囊性纤维化市场收入的主导地位,预计在 2024-2032 年预测期内,口服药物将成为增长最快的细分市场。这是因为这些药物用于治疗 CF 的使用有所增加。这些药物目前常用于治疗囊性纤维化 (CF),其他新型 CFTR 药物正在接受临床研究。这些药物的主要目标是延长患者的生命。
囊性纤维化区域洞察
按地区划分,本研究提供了北美、欧洲、亚太地区和世界其他地区的市场洞察。北美囊性纤维化市场在 2022 年的市场规模为 24 亿美元,占比约为 45.80%,预计在研究期间将呈现显著的复合年增长率。由于囊性纤维化患病率的上升和可用的治疗方法种类繁多,该地区对本研究的贡献最大。区域市场扩张将受到 CF 药物研发资金增加以及其他有益的政府举措的推动。
此外,市场报告中研究的主要国家是:美国、加拿大、德国、法国、英国、意大利、西班牙、中国、日本、印度、澳大利亚、韩国和巴西。
图 3:2022 年各地区囊性纤维化市场份额(%)
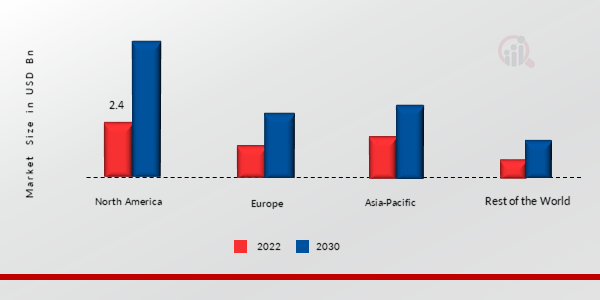
来源:二手研究、一手研究、MRFR 数据库和分析师评论
亚太囊性囊性纤维化市场占据第二大市场份额。随着城市生活方式的普及,该地区的患者数量也在增长。这可能会推动亚太地区市场以及医疗保健支出的增加。此外,中国囊性纤维化市场占有最大的市场份额,而印度囊性纤维化市场是该地区增长最快的市场。
由于囊性纤维化患病率的上升,预计欧洲囊性纤维化市场将在 2024 年至 2032 年期间以最快的复合年增长率增长。政府对创新的支持不断增加以及对各种 CF 药物研发计划的资金增加进一步推动了该地区的市场扩张。由于该地区囊性纤维化发病率的上升,英国贡献的资金最多。此外,英国囊性纤维化市场占有最大的市场份额,而德国囊性纤维化市场是该地区增长最快的市场。
囊性纤维化主要市场参与者和竞争洞察
主要市场参与者正在投入大量资金进行研发,以扩大其产品线,这将有助于囊性纤维化市场进一步增长。市场参与者还采取了一系列战略举措,以扩大其全球影响力,关键的市场发展包括新产品发布、合同协议、并购、增加投资以及与其他组织的合作。囊性纤维化行业的竞争对手必须提供具有成本效益的产品,才能在竞争日益激烈、蓬勃发展的市场环境中扩张并生存下来。
全球囊性纤维化行业制造商为使客户受益并扩大市场而采用的主要商业策略之一是本地化生产以降低运营成本。近年来,囊性纤维化行业为医学带来了一些最显著的益处。囊性纤维化市场的主要参与者包括 Allergan PLC、Chiesi Farmaceutici SpA、AbbVie、Hoffmann-La Roche AG、Gilead Sciences、PTC Therapeutics、Novartis AG、Vertex Pharmaceuticals Incorporated 和 Pharmaxis Ltd.
Roche 是 F. Hoffmann-La Roche AG 的品牌名称,这是一家全球医疗保健集团,拥有两个部门:制药和诊断。其控股公司 Roche Holding AG 的股票在瑞士证券交易所交易。公司总部位于巴塞尔。 2020年10月,罗氏宣布与AbbVie合作并收购其TMEM16A增效剂产品组合,这两种产品都将用于治疗囊性纤维化患者(无论其CFTR基因型如何)以及其他呼吸系统疾病患者。该协议将影响公司扩大产品供应的能力。
美国生物制药公司艾伯维(AbbVie)成立于2013年,是一家上市公司。它最初是雅培实验室(Abbott Laboratories)的分拆公司。 2019 年 10 月,艾伯维公司 (AbbVie Inc.) 与囊性纤维化基金会 (Cystic Fibrosis Foundation) 共同开发了囊性纤维化跨膜电导调节剂 (CFTR) 增效剂化学品。
囊性纤维化市场的主要公司包括
p
囊性纤维化行业发展
2020 年 10 月: 为了治疗囊性纤维化患者(无论 CFTR 基因型和其他呼吸系统疾病),罗氏宣布与 AbbVie 合作并收购 TMEM16A 增效剂产品组合。
2019 年 4 月: Vertex Pharmaceuticals Incorporated 的 KALYDECO(ivacaftor)药物已获得美国食品药品监督管理局 (FDA) 的批准,旨在为数百万患有囊性纤维化 (CF) 的年轻人增加治疗选择。由于有可能改变疾病的进程,这使得医生能够开始治疗符合条件的婴儿(最小六个月大)的囊性纤维化潜在病因。
囊性纤维化市场细分
h3
囊性纤维化治疗方法展望(十亿美元,2018-2030 年) ul
h3
囊性纤维化给药途径展望(十亿美元,2018-2030 年) ul
h3
囊性纤维化药物类别展望(十亿美元, 2018-2030) ul
p
囊性纤维化区域展望(十亿美元,2018-2030)















